Arctogalidia trivirgata
The small-toothed palm civet also known as the three-striped palm civet.
The small-toothed palm civet also known as the three-striped palm civet.
OUTR : 3-2-2024
Viverridae is a family of small to medium-sized, feliform mammals. The viverrids comprise 33 species placed in 14 genera. This family was named and first described by John Edward Gray in 1821.
Viverrids occur all over Africa, southern Europe, and South and Southeast Asia, across the Wallace Line.
Almost all viverrids outside the subfamily Genettinae are commonly called civets, but some civets are not viverrids.
Animals of the subfamily Genettinae are known as genets and oyans.
The word viverridae comes from the Latin viverra 'ferret', but ferrets are in a different family, the Mustelidae.
Viverrids have four or five toes on each foot and half-retractile claws. They have six incisors in each jaw and molars with two tubercular grinders behind in the upper jaw, and one in the lower jaw. The tongue is rough with sharp prickles. A pouch or gland occurs beneath the anus, but there is no cecum.
Viverrids are the most primitive of all the families of feliform Carnivora and clearly less specialized than the Felidae.
In external characteristics, they are distinguished from the Felidae by the longer muzzle and tuft of facial vibrissae between the lower jaw bones, and by the shorter limbs and the five-toed hind foot with the first digit present.
The skull differs by the position of the postpalatine foramina on the maxilla, almost always well in advance of the maxillopalatine suture, and usually about the level of the second premolar, and by the distinct external division of the auditory bulla into its two elements either by a definite groove or when rarely this is obliterated, by the depression of the tympanic bone in front of the swollen entotympanic.
The typical dental formula is: 3.1.4.2 / 3.1.4.2, but the number may be reduced, although never to the same extent as in the Felidae.
Their flesh-shearing carnassial teeth are relatively undeveloped compared to those of other feliform carnivorans. Most viverrid species have a penis bone (a baculum).
Arctogalidia trivirgata, The small-toothed palm civet also known as the three-striped palm civet, is a viverrid native to dense forests of Southeast Asia, from the Assam district of India to Indochina and the Malay Peninsula and on Sumatra, Bangka, Java, Borneo, and numerous small nearby islands of Indonesia.
The first scientific description by John Edward Gray in 1832 was based on a zoological specimen from the Maluku Islands in the collection of the Rijksmuseum van Natuurlijke Historie in Leiden, Netherlands.
It is blackish grey, has black paws and three black longitudinal stripes on the back.
A monotypic genus, Arctogalidia means ‘bear-weasel’ (from ancient Greek arkto- ‘bear’ + galidia ‘little weasel’). The specific epithet trivirgata means ‘three-striped’ in Latin.
The small-toothed palm civet is mid-sized by the standards of its family, weighing 2.4 kg (5.3 lb) and measuring 53 cm (21 in) long along the body, plus a tail of 58 cm (23 in). It has short fur that is generally a tawny or buff color while the head is a darker greyish tawny. Its muzzle is brown with a white streak that extends from the nose to the forehead. Only the females have the perineal scent gland, located near the vulva.
The diet is varied and omnivorous, and usually consists of insects, small mammals, nesting birds, fruits, frogs and lizards.
Matching the habits of other palm civets, this species is solitary, arboreal and nocturnal. Its gestation period is 45 days, and the average litter size is 3, which are born in dens made in the trees.
Young open their eyes at 11 days and are weaned at two months. It can have two litters a year and there is no set mating season. It can live for 11 years. It is threatened primarily by deforestation, as are many Southeast Asian forest animals.
Phylum : Chordata
Class : Mammalia
Order : Carnivora
Suborder : Feliformia
Family : Viverridae
Subfamily : Paradoxurinae
Genus : Arctogalidia
Merriam, 1897
Species : A. trivirgata
Binomial name Arctogalidia trivirgata
(Gray, 1832)
Viverrids occur all over Africa, southern Europe, and South and Southeast Asia, across the Wallace Line.
Almost all viverrids outside the subfamily Genettinae are commonly called civets, but some civets are not viverrids.
Animals of the subfamily Genettinae are known as genets and oyans.
The word viverridae comes from the Latin viverra 'ferret', but ferrets are in a different family, the Mustelidae.
Viverrids have four or five toes on each foot and half-retractile claws. They have six incisors in each jaw and molars with two tubercular grinders behind in the upper jaw, and one in the lower jaw. The tongue is rough with sharp prickles. A pouch or gland occurs beneath the anus, but there is no cecum.
Viverrids are the most primitive of all the families of feliform Carnivora and clearly less specialized than the Felidae.
In external characteristics, they are distinguished from the Felidae by the longer muzzle and tuft of facial vibrissae between the lower jaw bones, and by the shorter limbs and the five-toed hind foot with the first digit present.
The skull differs by the position of the postpalatine foramina on the maxilla, almost always well in advance of the maxillopalatine suture, and usually about the level of the second premolar, and by the distinct external division of the auditory bulla into its two elements either by a definite groove or when rarely this is obliterated, by the depression of the tympanic bone in front of the swollen entotympanic.
The typical dental formula is: 3.1.4.2 / 3.1.4.2, but the number may be reduced, although never to the same extent as in the Felidae.
Their flesh-shearing carnassial teeth are relatively undeveloped compared to those of other feliform carnivorans. Most viverrid species have a penis bone (a baculum).
Arctogalidia trivirgata, The small-toothed palm civet also known as the three-striped palm civet, is a viverrid native to dense forests of Southeast Asia, from the Assam district of India to Indochina and the Malay Peninsula and on Sumatra, Bangka, Java, Borneo, and numerous small nearby islands of Indonesia.
The first scientific description by John Edward Gray in 1832 was based on a zoological specimen from the Maluku Islands in the collection of the Rijksmuseum van Natuurlijke Historie in Leiden, Netherlands.
It is blackish grey, has black paws and three black longitudinal stripes on the back.
A monotypic genus, Arctogalidia means ‘bear-weasel’ (from ancient Greek arkto- ‘bear’ + galidia ‘little weasel’). The specific epithet trivirgata means ‘three-striped’ in Latin.
The small-toothed palm civet is mid-sized by the standards of its family, weighing 2.4 kg (5.3 lb) and measuring 53 cm (21 in) long along the body, plus a tail of 58 cm (23 in). It has short fur that is generally a tawny or buff color while the head is a darker greyish tawny. Its muzzle is brown with a white streak that extends from the nose to the forehead. Only the females have the perineal scent gland, located near the vulva.
The diet is varied and omnivorous, and usually consists of insects, small mammals, nesting birds, fruits, frogs and lizards.
Matching the habits of other palm civets, this species is solitary, arboreal and nocturnal. Its gestation period is 45 days, and the average litter size is 3, which are born in dens made in the trees.
Young open their eyes at 11 days and are weaned at two months. It can have two litters a year and there is no set mating season. It can live for 11 years. It is threatened primarily by deforestation, as are many Southeast Asian forest animals.
Phylum : Chordata
Class : Mammalia
Order : Carnivora
Suborder : Feliformia
Family : Viverridae
Subfamily : Paradoxurinae
Genus : Arctogalidia
Merriam, 1897
Species : A. trivirgata
Binomial name Arctogalidia trivirgata
(Gray, 1832)
Callosciurus finlaysonii
leucistic squirrels
leucistic squirrels
Dec 2023 : Sakol Nakhon village temple ground
Tree squirrels are the members of the squirrel family (Sciuridae) commonly just referred to as "squirrels". They include more than 100 arboreal species native to all continents except Antarctica and Oceania.
They do not form a single natural, or monophyletic, group, they are variously related to others in the squirrel family, including ground squirrels, flying squirrels, marmots, and chipmunks.
The defining characteristic used to determine which species of Sciuridae are tree squirrels is dependent on their habitat rather than their physiology.
Tree squirrels live mostly among trees, as opposed to those that live in burrows in the ground or among rocks. An exception is the flying squirrel that also makes its home in trees, but has a physiological distinction separating it from its tree squirrel cousins: special flaps of skin called patagia, acting as glider wings, which allow gliding flight.
Squirrels come in a variety of different colors. So, what causes white squirrels and just how rare are they in the world today?
There are two basic types of white squirrels: albino and leucistic. Albinos have pink or blue eyes and no dark pigments anywhere on their bodies.
Squirrels that have white coats and dark eyes are called leucistic. Another type of white squirrel is predominantly white and sports a dark head patch and stripe on its back.
White squirrels are not a species of their own such as fox squirrels or the Mexican gray squirrel. Instead, white squirrels are most often members of the eastern gray squirrel species, Sciurus carolinensis.
A white squirrel is not merely a squirrel that has white integrated into its fur colors. Instead, these creatures are unusually white for a few different reasons.
One option is that squirrels could have greatly reduced pigmentation in their fur due to leucism. As a result, they have all white fur except for the dorsal stripe of gray fur that runs down the length of their bodies.
Albino gray squirrels are the rarest form of white squirrel. Mammalogists estimate that the odds of a female gray squirrel giving birth to an albino offspring are 1 in 100,000.
The scarcity of albino squirrels is thought to be at least partially linked to the animal’s lack of any dark eye pigment: This leaves them more susceptible to the harmful effects of sun rays, which can permanently damage their eyes. In turn, their poor eyesight makes them more likely to suffer injury and death by falling out of trees.
To be clear, white squirrels are not albino. Albino squirrels are very different because albinism impacts their melanin production. As a result, they have red or pink eyes in most cases. When most people talk about white squirrels, they are referring to the leucistic ones and not the albino ones.
White squirrels exist due to leucism in existing squirrel species. Leucism is when an animal has a partial loss of pigmentation. Most mammals can be leucistic.
In this case, leucism comes as a result of a mutated gene in the animals causing them to have greatly reduced pigmentation in their fur but not changes in other parts of their body like that seen with albinism.
The expression of this recessive allele is very rare. In fact, it’s much rarer to see a white squirrel than it is to see a melanistic black squirrel.
In North America, the eastern gray squirrel is one of the species that most often appears white. White squirrels are not a natural occurrence in the sense that no squirrel species are entirely white, at least in North America. Some tree squirrels in Thailand are white.
White squirrels are not even entirely white. They often have a gray patch on the crown of their heads along with a gray stripe on the top of their bodies.
Finlayson's squirrel or the variable squirrel (Callosciurus finlaysonii, leucistic squirrels sometimes misspelled C. finlaysoni) is a species of rodent in the family Sciuridae. It is found in Cambodia, Laos, Myanmar, Thailand, and Vietnam.
The species occurs in a wide range of wooded habitats, including gardens and parks in cities like Bangkok. It was named in honour of the Scottish naturalist and traveller George Finlayson.
Phylum : Chordata
Class : Mammalia
Order : Rodentia
Suborder : Sciuromorpha
Family : Sciuridae
Fischer de Waldheim, 1817
Genus : Callosciurus
Species : C. finlaysonii
Binomial name Callosciurus finlaysonii
(Horsfield, 1823)
They do not form a single natural, or monophyletic, group, they are variously related to others in the squirrel family, including ground squirrels, flying squirrels, marmots, and chipmunks.
The defining characteristic used to determine which species of Sciuridae are tree squirrels is dependent on their habitat rather than their physiology.
Tree squirrels live mostly among trees, as opposed to those that live in burrows in the ground or among rocks. An exception is the flying squirrel that also makes its home in trees, but has a physiological distinction separating it from its tree squirrel cousins: special flaps of skin called patagia, acting as glider wings, which allow gliding flight.
Squirrels come in a variety of different colors. So, what causes white squirrels and just how rare are they in the world today?
There are two basic types of white squirrels: albino and leucistic. Albinos have pink or blue eyes and no dark pigments anywhere on their bodies.
Squirrels that have white coats and dark eyes are called leucistic. Another type of white squirrel is predominantly white and sports a dark head patch and stripe on its back.
White squirrels are not a species of their own such as fox squirrels or the Mexican gray squirrel. Instead, white squirrels are most often members of the eastern gray squirrel species, Sciurus carolinensis.
A white squirrel is not merely a squirrel that has white integrated into its fur colors. Instead, these creatures are unusually white for a few different reasons.
One option is that squirrels could have greatly reduced pigmentation in their fur due to leucism. As a result, they have all white fur except for the dorsal stripe of gray fur that runs down the length of their bodies.
Albino gray squirrels are the rarest form of white squirrel. Mammalogists estimate that the odds of a female gray squirrel giving birth to an albino offspring are 1 in 100,000.
The scarcity of albino squirrels is thought to be at least partially linked to the animal’s lack of any dark eye pigment: This leaves them more susceptible to the harmful effects of sun rays, which can permanently damage their eyes. In turn, their poor eyesight makes them more likely to suffer injury and death by falling out of trees.
To be clear, white squirrels are not albino. Albino squirrels are very different because albinism impacts their melanin production. As a result, they have red or pink eyes in most cases. When most people talk about white squirrels, they are referring to the leucistic ones and not the albino ones.
White squirrels exist due to leucism in existing squirrel species. Leucism is when an animal has a partial loss of pigmentation. Most mammals can be leucistic.
In this case, leucism comes as a result of a mutated gene in the animals causing them to have greatly reduced pigmentation in their fur but not changes in other parts of their body like that seen with albinism.
The expression of this recessive allele is very rare. In fact, it’s much rarer to see a white squirrel than it is to see a melanistic black squirrel.
In North America, the eastern gray squirrel is one of the species that most often appears white. White squirrels are not a natural occurrence in the sense that no squirrel species are entirely white, at least in North America. Some tree squirrels in Thailand are white.
White squirrels are not even entirely white. They often have a gray patch on the crown of their heads along with a gray stripe on the top of their bodies.
Finlayson's squirrel or the variable squirrel (Callosciurus finlaysonii, leucistic squirrels sometimes misspelled C. finlaysoni) is a species of rodent in the family Sciuridae. It is found in Cambodia, Laos, Myanmar, Thailand, and Vietnam.
The species occurs in a wide range of wooded habitats, including gardens and parks in cities like Bangkok. It was named in honour of the Scottish naturalist and traveller George Finlayson.
Phylum : Chordata
Class : Mammalia
Order : Rodentia
Suborder : Sciuromorpha
Family : Sciuridae
Fischer de Waldheim, 1817
Genus : Callosciurus
Species : C. finlaysonii
Binomial name Callosciurus finlaysonii
(Horsfield, 1823)
Tragulus napu
greater mouse deer
greater mouse deer
Pulau ubin : 5-11-23
Tragulus is a genus of even-toed ungulates in the family Tragulidae that are known as mouse-deer.
In Ancient Greek τράγος (tragos) means a male goat, while the Latin diminutive –ulus means 'tiny'. With a weight of 0.7–8.0 kg (1.5–17.6 lb) and a length of 40–75 cm (16–30 in), they are the smallest ungulates in the world, though the largest species of mouse-deer surpass some species of Neotragus antelopes in size.
The mouse-deer are restricted to Southeast Asia from far southern China (south Yunnan) to the Philippines (Balabac) and Java.
Following recent taxonomic changes, several of the species in this genus are poorly known, but all are believed to be mainly nocturnal and feed on leaves, fruits, grasses, and other vegetation in the dense forest undergrowth.
They are solitary or live in pairs, and the males have elongated canine teeth (neither gender has horns or antlers) that are used in fights. Unlike other members of their family, the Tragulus mouse-deer lack obvious pale stripes/spots on their upper parts.
Traditionally, only two species of mouse-deer in the genus Tragulus have been recognized: The relatively large T. napu and the small T. javanicus.
Following a review in 2004, T. nigricans and T. versicolor were split from T. napu, and T. kanchil and T. williamsoni were split from T. javanicus. With these changes, T. kanchil and T. napu are the most widespread species, while the remaining have far smaller distributions (though some uncertainty over the exact distribution limits of the various species in Indochina remain).
The greater mouse-deer, greater Malay chevrotain, or napu (Tragulus napu) is a species of even-toed ungulate in the family Tragulidae found in Sumatra, Borneo, and smaller Malaysian and Indonesian
islands, and in southern Myanmar, southern Thailand and peninsular Malaysia. Its natural habitat is subtropical or tropical, moist, lowland forest.
The greater mouse-deer is an even-toed ungulate. Although very small for an ungulate, the greater mouse-deer is one of the largest members of its genus. It is rivalled in size by Williamson's mouse-deer.
It weighs 5 to 8 kg (11 to 18 lb). Its head-and-body length is 70 to 75 cm (2.30 to 2.46 ft) and its tail length is 8 to 10 cm (3.1 to 3.9 in). Its shoulder height is about 30 to 35 cm (12 to 14 in) It has a small, triangular head with a small, pointed, black nose and large eyes. Its long legs are as thin as a pencil.
The hind legs are visibly longer than the front legs. The body is rounded. The fur on the upper part of its body is grey-buff to orange-buff. On the sides, the fur is quite pale, but darker along the midline. It is white underneath, more specifically on the neck, stomach, chest, and chin. The male has neither horns nor antlers, but has small "tusks" – elongated canines in the upper jaws.
Thought to be regionally extinct in Singapore, they were rediscovered on an offshore island in 2008. Reports of its occurrence elsewhere are probably incorrect. They are terrestrial, but spend time in wet, swampy areas.
The greater mouse-deer is solitary and nocturnal. It uses small trails through thick brush in the forest.
When the male is ready to mate, he rubs a large gland on his lower jaw against the female to determine whether she is ready to mate. If she is not ready, she responds by walking away. The male is very territorial, marking his territory with feces, urine, and secretions from the intermandibular gland under the chin. When angry, the male beats the ground with his hooves at a rate of four times per second.
They are rather trusting but delicate animals. They feed on fallen fruits, aquatic plants, buds, leaves, shrubs and grasses.
Greater mouse-deer breed throughout the year, the female spends most of her adult life pregnant. They usually produce one young per birth, after a gestation of 152–155 days. Newborn animals are well-developed and immediately able to stand, they are fully active after 30 minutes. The young stand on three legs while nursing. Both male and female become mature at age 4½ months. Their lifespan is up to 14 years.
The major threats to T. napu are overhunting by humans and loss of habitat through rapid
deforestation.
Phylum : Chordata
Class : Mammalia
Order : Artiodactyla
Family : Tragulidae
Genus :Tragulus
Species: T. napu
Binomial name Tragulus napu
(F. Cuvier, 1822)
In Ancient Greek τράγος (tragos) means a male goat, while the Latin diminutive –ulus means 'tiny'. With a weight of 0.7–8.0 kg (1.5–17.6 lb) and a length of 40–75 cm (16–30 in), they are the smallest ungulates in the world, though the largest species of mouse-deer surpass some species of Neotragus antelopes in size.
The mouse-deer are restricted to Southeast Asia from far southern China (south Yunnan) to the Philippines (Balabac) and Java.
Following recent taxonomic changes, several of the species in this genus are poorly known, but all are believed to be mainly nocturnal and feed on leaves, fruits, grasses, and other vegetation in the dense forest undergrowth.
They are solitary or live in pairs, and the males have elongated canine teeth (neither gender has horns or antlers) that are used in fights. Unlike other members of their family, the Tragulus mouse-deer lack obvious pale stripes/spots on their upper parts.
Traditionally, only two species of mouse-deer in the genus Tragulus have been recognized: The relatively large T. napu and the small T. javanicus.
Following a review in 2004, T. nigricans and T. versicolor were split from T. napu, and T. kanchil and T. williamsoni were split from T. javanicus. With these changes, T. kanchil and T. napu are the most widespread species, while the remaining have far smaller distributions (though some uncertainty over the exact distribution limits of the various species in Indochina remain).
The greater mouse-deer, greater Malay chevrotain, or napu (Tragulus napu) is a species of even-toed ungulate in the family Tragulidae found in Sumatra, Borneo, and smaller Malaysian and Indonesian
islands, and in southern Myanmar, southern Thailand and peninsular Malaysia. Its natural habitat is subtropical or tropical, moist, lowland forest.
The greater mouse-deer is an even-toed ungulate. Although very small for an ungulate, the greater mouse-deer is one of the largest members of its genus. It is rivalled in size by Williamson's mouse-deer.
It weighs 5 to 8 kg (11 to 18 lb). Its head-and-body length is 70 to 75 cm (2.30 to 2.46 ft) and its tail length is 8 to 10 cm (3.1 to 3.9 in). Its shoulder height is about 30 to 35 cm (12 to 14 in) It has a small, triangular head with a small, pointed, black nose and large eyes. Its long legs are as thin as a pencil.
The hind legs are visibly longer than the front legs. The body is rounded. The fur on the upper part of its body is grey-buff to orange-buff. On the sides, the fur is quite pale, but darker along the midline. It is white underneath, more specifically on the neck, stomach, chest, and chin. The male has neither horns nor antlers, but has small "tusks" – elongated canines in the upper jaws.
Thought to be regionally extinct in Singapore, they were rediscovered on an offshore island in 2008. Reports of its occurrence elsewhere are probably incorrect. They are terrestrial, but spend time in wet, swampy areas.
The greater mouse-deer is solitary and nocturnal. It uses small trails through thick brush in the forest.
When the male is ready to mate, he rubs a large gland on his lower jaw against the female to determine whether she is ready to mate. If she is not ready, she responds by walking away. The male is very territorial, marking his territory with feces, urine, and secretions from the intermandibular gland under the chin. When angry, the male beats the ground with his hooves at a rate of four times per second.
They are rather trusting but delicate animals. They feed on fallen fruits, aquatic plants, buds, leaves, shrubs and grasses.
Greater mouse-deer breed throughout the year, the female spends most of her adult life pregnant. They usually produce one young per birth, after a gestation of 152–155 days. Newborn animals are well-developed and immediately able to stand, they are fully active after 30 minutes. The young stand on three legs while nursing. Both male and female become mature at age 4½ months. Their lifespan is up to 14 years.
The major threats to T. napu are overhunting by humans and loss of habitat through rapid
deforestation.
Phylum : Chordata
Class : Mammalia
Order : Artiodactyla
Family : Tragulidae
Genus :Tragulus
Species: T. napu
Binomial name Tragulus napu
(F. Cuvier, 1822)
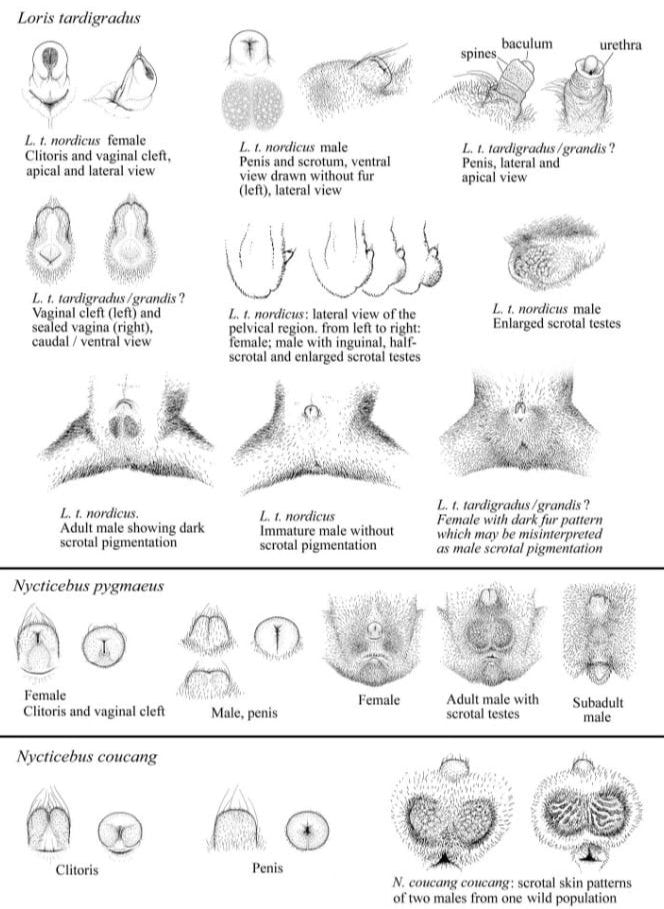
Nycticebus coucang
Sunda slow loris
Sunda slow loris
The Sunda slow loris (Nycticebus coucang) or greater slow loris is a strepsirrhine primate and a species of slow loris native to Indonesia, West Malaysia,
southern Thailand and Singapore. The species is generally solitary.
Slow lorises (genus Nycticebus) are strepsirrhine primates and are related to other living lorisoids, such as the pygmy slow loris (Xanthonycticebus), slender
lorises (Loris), pottos (Perodicticus), false pottos (Pseudopotto), angwantibos (Arctocebus), and galagos (family Galagidae), and to the lemurs of Madagascar.
They are most closely related to the pygmy slow loris, followed by the slender lorises of South Asia, the angwantibos, pottos and false pottos of Central and West Africa.
Lorisoids are thought to have evolved in Africa, where most living species occur, later, one group may have migrated to Asia and evolved into the slender and slow lorises of today.
The Sunda slow loris is nocturnal and arboreal,
typically occurring in evergreen forests. It prefers rainforests with continuous dense canopies and
has an extremely low metabolic rate compared to other mammals of its size. Slow lorises move slowly and deliberately, making little or no noise, and when threatened, they stop moving and remain motionless.
Slow lorises have stout bodies, it measures 27 to 38 cm (11 to 15 in) from head to tail and weighs between 599 and 685 g (21.1 and 24.2 oz) it has a wet nose
(rhinarium), a round head, small ears hidden in thick fur, a flat face, large eyes and a vestigial tail.
The limbs are thick and short, with nails on the fingers and toes, only the second toe of the hind limb has claws, and the second finger of the forelimb is degenerated, leaving only a short one refer to.
The ears are small, sparsely covered in hair, and hidden in the fur. Similar to the slender lorises, the fur around and directly above the eyes is dark. Unlike the slender lorises, however, the white stripe that separates the eye rings broadens both on the tip of the nose and on the forehead while also fading out on the forehead. Like other strepsirrhine primates, the nose and lip are covered by a moist skin called the rhinarium ("wet nose"), which is a sense organ.
The eyes of slow lorises are forward-facing, which gives stereo vision. Their eyes are large and possess a reflective layer, called the tapetum lucidum, that improves low-light vision to help them see better at night. It is possible that this layer blurs the images they see, as the reflected light may interfere with the incoming light.
Slow lorises have monochromatic vision, meaning they see in shades of only one color. They lack the
opsin gene that would allow them to detect short wavelength light, which includes the colors blue and green. It has strong grasping hands and feet with opposable thumbs and big toes, allowing it to grip tree branches very tightly.
N.coucang is Omnivore. Their diet consists of bird
eggs, small vertebrates, fruit, sap, floral nectar, and arthropods. They are also known to feed on molluscs, including the giant land snail Achatina fulica. It will
feed on exudates such as gum and sap by licking wounds in trees.
Slow lorises are the only venomous primate known to secrete a brown oil in their underarms when threatened, which mixes with saliva to create a poisonous substance (the oil and saliva are also toxic themselves) . Toxins are obtained by eating poisonous insects, such as centipedes and scorpions.
Slow lorises have a toxic bite due to a toxin that is produced by licking a gland on their inner elbow, the brachial organ. Saliva from the slow loris is required to activate the secretion from the arm gland. However, very little else is known about the chemical nature of the toxin.
The slow loris bite is reported to be painful in humans with symptoms including burning of the tongue and throat, hypotension, muscle convulsions, heart and respiratory problems, unconsciousness, and even death through anaphylaxis shock.
Treatment :
Vaccination against tetanus and antibiotics is often given to bite victims. Treatment is symptomatic for bites and anaphylaxis.
Though N.coucang are mainly known to be solitary it may fit into the monogamous single male / single female social system with the offspring living with the parents.
It sleeps during the day, rolled up in a ball in hidden parts of trees above the ground, often on branches, twigs, palm fronds, or lianas.
The species is polyoestrous, usually giving birth to a single offspring after a gestation period of 192 days. The young disperses between 16 and 27 months, generally when it is sexually mature.
A slow loris is born with its eyes open and already has the strength and ability to hold onto branches. Following birth, it will stay with its mother everyday for about 7 weeks.
The baby slow loris clings to the stomach of the mother wherever she goes, except for when she is searching for food. When the mother goes out in search of food, she cleans her babies first. This is not a normal cleaning though, venom from the mother
mixes with saliva and gets spread on the young, which will keep them safe if a predator finds them.
It will nurse for about 6 months, but it is able to eat solid food within 4 weeks following birth. Infants grow and can crawl through the trees by the age of 2 weeks.
In less that a year, an adolescent slow loris is able to fend for itself and has not developed any sexual dimorphism (differences between genders) by weight.
As a loris grows older, it can show some sexual dimorphism with males being slightly heavier than the females. On average, Nycticebus coucang grow to be about 15 inches long, have 1 inch vestigial tails, and can weight about 3 pounds. They have the ability to live to 20 years of age, and can slightly exceed that age in captivity.
The species is listed as endangered on the IUCN Red List. It is threatened with extinction due to a growing demand in the exotic pet trade, and has become one of the most abundant primate species on sale at Indonesian pet markets.
The common name, Sunda slow loris, refers to the Sunda Islands, a group of islands in the western part of the Malay archipelago where it is found. Another common name for the species is the greater slow loris.
The specific name, coucang, derives from kukang, its common name in Indonesia. It is commonly known as malu-malu, meaning "shy" in Indonesian, and also as bukang or Kalamasan. It is sometimes called kuskus, because local people do not distinguish between the slow loris and cuscus which belong to a group of Australasian possums.
In Malaysia they are sometimes known as kongkang or kera duku, kera is Malay for monkey while duku is the
fruit-bearing tree, Lansium parasiticum.
In Thailand, it is called ling lom (ลิงลม), which
translates as "wind monkey".
The Sunda slow loris has dark rings around its large eyes, a white nose with a whitish strip that extends to the forehead and a dark stripe that stretches from the back of the head along the spine.
Its soft, thick, woolly fur ranges from light brown to deep reddish brown, with a lighter underside.
The species is distinct from the Bengal slow loris due to the dark inverse teardrop markings around the eyes which meet the dark dorsal stripe on the back
of the head. It tends to have a much more distinct white stripe between the eyes, more distinct dark coloring around the eyes, and a browner coat than the Bengal slow loris which is larger, grayer, and shows less contrast.
The Sunda slow loris has less white facial coloring than the much smaller pygmy slow loris. Local color variations are known to occur.
The vestigial tail, hidden beneath the fur, is reduced to a stump. It has a toothcomb, six forward-facing teeth on the bottom jaw, which includes the lower incisors and the canine teeth. The structure is generally used for grooming in other strepsirhine primates, but lorises also use it to scrape off gum when foraging.
It has a shortened second digit, and the hands have a strong grip. Like other lorises, it excretes a strong
lsmelling liquid from glands beneath its arms which is used in communication.
One major distinguishing feature between all loris species is locomotion, the Sunda slow loris moves slowly through trees on all four limbs, typically with three limbs attached to a support at a time.
Its movement has been described as unique; similar to crawling, or as if it was climbing in every direction, the Sunda slow loris changes direction or moves between branches with little noise or change in speed.
In captivity, about a quarter of its time is spent moving quadrupedally, a quarter suspended or hanging, a quarter climbing, and a quarter clasping multiple branches (bridging). It may hang below a branch by one or both feet for long periods of time.
The Sunda slow loris is an arboreal and nocturnal
primate, resting by day in the forks of trees, or in thick vegetation and feeding on fruit and insects by night, unlike other loris species, it remains in trees most of its life, while the Bengal slow loris will often sleep on the ground, the Sunda slow loris sleeps in a ball in branches or foliage.
It usually sleeps alone but has been observed to sleep with several conspecifics (individuals of the same species), including other adults.
Despite its slow metabolism rate, the Sunda slow loris has a high-energy diet. Its slow lifestyle may be due to the energy costs of detoxifying certain secondary plant compounds in many genera of food plants their diets.
All slow loris species produce a toxin in glands on the insides of their elbows. This is spread across their bodies and those of their offspring using the toothcomb while grooming. When threatened with predators, the Sunda slow loris can bite, roll into a ball exposing its toxic saliva-covered fur, or roll up and drop from the trees.
However, the primary method of predator avoidance is crypsis, whereby it hides. The Asiatic reticulated python, the changeable hawk-eagle and the Sumatran orangutan have been recorded as predators of the Sunda slow loris.
One study showed only 8% of the species' active time was spent near other individuals. Where home ranges overlap, spatial groups are formed. These groups consist of one male, one female and up to three younger individuals. The interactions between these individuals are largely friendly; they include allogrooming, following, pant-growling, and click-calling, although social behaviors only make up around 3% of the activity budget.
When it comes into contact with conspecifics from other home ranges there is usually no reaction as home ranges are not defended.
In captivity however, it can be aggressive with other individuals. Males have shown antagonistic behaviors such as attacks, pursuits, threats, assertion, fighting, and subordination. The fighting often results in serious injury. Despite this, they are known to be generally sociable in captivity, with allogrooming being the most common social behavior.
There are eight distinct call types made by Sunda slow loris adults, which can be divided into two categories, contact and contact-seeking calls such as whistles and short keckers (a social play and attention-seeking call), and aggressive and defensive calls such as long keckers, screams, snarls and grunts.
Because they rely on crypsis to avoid predators, they do not make alarm calls. Infants emits clicks and squeaks when disturbed. During estrus, females make whistle calls when in visual contact with a male. When exploring new environments and during handling, it makes ultrasonic vocalisations out of the human hearing range.
Olfactory communication is very important in this species. It has roles in alerting others of the identity, physical state and position of the individual. It is also important in reproduction.
Like other slow lorises, the Sunda slow loris has glands on its elbows that exude oils. The gland is licked to spread scent and is thought to have evolved for communication, but it is toxic to humans. If the person is allergic to the animal they can go into shock and could even die. It also has glands on its anus
and can signal conspecifics by perineal rubbing which deposits urine.
When it locates the scent of another slow loris it may rub its face on the substrate where the scent was found. The Sunda slow loris may grin or bare its teeth. When stressed, infants may grin, while adults bear their teeth to show aggression or fear, but also during play.
Reproduction is one of the few times the Sunda slow loris aggregates with conspecifics, as it is largely solitary. One study recorded that the most slow lorises ever seen together was six, this appeared to be a female in estrus and five males following her.
This may suggest a more promiscuous mating system, where females mate with more than one male. Despite this, its testis size is small compared with similar sized promsimians, which is indicative of monogamy. In the wild the mating system of the Sunda slow loris is thought to vary between populations.
Sexual maturity is reached between the ages of 18 and 24 months in females, and can be reached by 17 months in males. It is polyestrous, having many periods of sexual receptivity during a year.
In captivity however, there is a clear birth peak between March and May. It has been observed that reproductive patterns of captive prosimians in the northern hemisphere are altered. The estrus cycle lasts 29–45 days, with most copulations occurring on the same day.
Females in estrus are followed by males, with copulation initiated by the female. The female will hang from a branch and may vocalise. The male will hold the female and the branch and copulate with her. Urine-marking and vocalising are also used by the female to solicit mating. The male may create a mating plug following copulation.
The gestation period averages 192.2 days, after which one young is born, although twinning has been observed. Both males and females will disperse in the wild, this occurs between 16 and 27 months.
The Sunda slow loris is found in continuous canopy tropical rainforests. It is adaptable and will also live in other types of habitat. It is found in Indonesia, on the islands of Sumatra, Batam and Galang in the Riau Archipelago, and Tebing Tinggi Island and Great Natuna (Bunguran) in the Natuna Islands; in Malaysia on the Malay Peninsula and Pulau Tioman; in the southern peninsular of Thailand; and Singapore.
The Sunda slow loris is sympatric shares its range with the Bengal slow loris in Thailand and
hybridisation has occurred.
Phylum: Chordata
Class: Mammalia
Order: Primates
Suborder: Strepsirrhini
Family: Lorisidae
Genus: Nycticebus
Species: N. coucang
Binomial name Nycticebus coucang
(Boddaert, 1785)
southern Thailand and Singapore. The species is generally solitary.
Slow lorises (genus Nycticebus) are strepsirrhine primates and are related to other living lorisoids, such as the pygmy slow loris (Xanthonycticebus), slender
lorises (Loris), pottos (Perodicticus), false pottos (Pseudopotto), angwantibos (Arctocebus), and galagos (family Galagidae), and to the lemurs of Madagascar.
They are most closely related to the pygmy slow loris, followed by the slender lorises of South Asia, the angwantibos, pottos and false pottos of Central and West Africa.
Lorisoids are thought to have evolved in Africa, where most living species occur, later, one group may have migrated to Asia and evolved into the slender and slow lorises of today.
The Sunda slow loris is nocturnal and arboreal,
typically occurring in evergreen forests. It prefers rainforests with continuous dense canopies and
has an extremely low metabolic rate compared to other mammals of its size. Slow lorises move slowly and deliberately, making little or no noise, and when threatened, they stop moving and remain motionless.
Slow lorises have stout bodies, it measures 27 to 38 cm (11 to 15 in) from head to tail and weighs between 599 and 685 g (21.1 and 24.2 oz) it has a wet nose
(rhinarium), a round head, small ears hidden in thick fur, a flat face, large eyes and a vestigial tail.
The limbs are thick and short, with nails on the fingers and toes, only the second toe of the hind limb has claws, and the second finger of the forelimb is degenerated, leaving only a short one refer to.
The ears are small, sparsely covered in hair, and hidden in the fur. Similar to the slender lorises, the fur around and directly above the eyes is dark. Unlike the slender lorises, however, the white stripe that separates the eye rings broadens both on the tip of the nose and on the forehead while also fading out on the forehead. Like other strepsirrhine primates, the nose and lip are covered by a moist skin called the rhinarium ("wet nose"), which is a sense organ.
The eyes of slow lorises are forward-facing, which gives stereo vision. Their eyes are large and possess a reflective layer, called the tapetum lucidum, that improves low-light vision to help them see better at night. It is possible that this layer blurs the images they see, as the reflected light may interfere with the incoming light.
Slow lorises have monochromatic vision, meaning they see in shades of only one color. They lack the
opsin gene that would allow them to detect short wavelength light, which includes the colors blue and green. It has strong grasping hands and feet with opposable thumbs and big toes, allowing it to grip tree branches very tightly.
N.coucang is Omnivore. Their diet consists of bird
eggs, small vertebrates, fruit, sap, floral nectar, and arthropods. They are also known to feed on molluscs, including the giant land snail Achatina fulica. It will
feed on exudates such as gum and sap by licking wounds in trees.
Slow lorises are the only venomous primate known to secrete a brown oil in their underarms when threatened, which mixes with saliva to create a poisonous substance (the oil and saliva are also toxic themselves) . Toxins are obtained by eating poisonous insects, such as centipedes and scorpions.
Slow lorises have a toxic bite due to a toxin that is produced by licking a gland on their inner elbow, the brachial organ. Saliva from the slow loris is required to activate the secretion from the arm gland. However, very little else is known about the chemical nature of the toxin.
The slow loris bite is reported to be painful in humans with symptoms including burning of the tongue and throat, hypotension, muscle convulsions, heart and respiratory problems, unconsciousness, and even death through anaphylaxis shock.
Treatment :
Vaccination against tetanus and antibiotics is often given to bite victims. Treatment is symptomatic for bites and anaphylaxis.
Though N.coucang are mainly known to be solitary it may fit into the monogamous single male / single female social system with the offspring living with the parents.
It sleeps during the day, rolled up in a ball in hidden parts of trees above the ground, often on branches, twigs, palm fronds, or lianas.
The species is polyoestrous, usually giving birth to a single offspring after a gestation period of 192 days. The young disperses between 16 and 27 months, generally when it is sexually mature.
A slow loris is born with its eyes open and already has the strength and ability to hold onto branches. Following birth, it will stay with its mother everyday for about 7 weeks.
The baby slow loris clings to the stomach of the mother wherever she goes, except for when she is searching for food. When the mother goes out in search of food, she cleans her babies first. This is not a normal cleaning though, venom from the mother
mixes with saliva and gets spread on the young, which will keep them safe if a predator finds them.
It will nurse for about 6 months, but it is able to eat solid food within 4 weeks following birth. Infants grow and can crawl through the trees by the age of 2 weeks.
In less that a year, an adolescent slow loris is able to fend for itself and has not developed any sexual dimorphism (differences between genders) by weight.
As a loris grows older, it can show some sexual dimorphism with males being slightly heavier than the females. On average, Nycticebus coucang grow to be about 15 inches long, have 1 inch vestigial tails, and can weight about 3 pounds. They have the ability to live to 20 years of age, and can slightly exceed that age in captivity.
The species is listed as endangered on the IUCN Red List. It is threatened with extinction due to a growing demand in the exotic pet trade, and has become one of the most abundant primate species on sale at Indonesian pet markets.
The common name, Sunda slow loris, refers to the Sunda Islands, a group of islands in the western part of the Malay archipelago where it is found. Another common name for the species is the greater slow loris.
The specific name, coucang, derives from kukang, its common name in Indonesia. It is commonly known as malu-malu, meaning "shy" in Indonesian, and also as bukang or Kalamasan. It is sometimes called kuskus, because local people do not distinguish between the slow loris and cuscus which belong to a group of Australasian possums.
In Malaysia they are sometimes known as kongkang or kera duku, kera is Malay for monkey while duku is the
fruit-bearing tree, Lansium parasiticum.
In Thailand, it is called ling lom (ลิงลม), which
translates as "wind monkey".
The Sunda slow loris has dark rings around its large eyes, a white nose with a whitish strip that extends to the forehead and a dark stripe that stretches from the back of the head along the spine.
Its soft, thick, woolly fur ranges from light brown to deep reddish brown, with a lighter underside.
The species is distinct from the Bengal slow loris due to the dark inverse teardrop markings around the eyes which meet the dark dorsal stripe on the back
of the head. It tends to have a much more distinct white stripe between the eyes, more distinct dark coloring around the eyes, and a browner coat than the Bengal slow loris which is larger, grayer, and shows less contrast.
The Sunda slow loris has less white facial coloring than the much smaller pygmy slow loris. Local color variations are known to occur.
The vestigial tail, hidden beneath the fur, is reduced to a stump. It has a toothcomb, six forward-facing teeth on the bottom jaw, which includes the lower incisors and the canine teeth. The structure is generally used for grooming in other strepsirhine primates, but lorises also use it to scrape off gum when foraging.
It has a shortened second digit, and the hands have a strong grip. Like other lorises, it excretes a strong
lsmelling liquid from glands beneath its arms which is used in communication.
One major distinguishing feature between all loris species is locomotion, the Sunda slow loris moves slowly through trees on all four limbs, typically with three limbs attached to a support at a time.
Its movement has been described as unique; similar to crawling, or as if it was climbing in every direction, the Sunda slow loris changes direction or moves between branches with little noise or change in speed.
In captivity, about a quarter of its time is spent moving quadrupedally, a quarter suspended or hanging, a quarter climbing, and a quarter clasping multiple branches (bridging). It may hang below a branch by one or both feet for long periods of time.
The Sunda slow loris is an arboreal and nocturnal
primate, resting by day in the forks of trees, or in thick vegetation and feeding on fruit and insects by night, unlike other loris species, it remains in trees most of its life, while the Bengal slow loris will often sleep on the ground, the Sunda slow loris sleeps in a ball in branches or foliage.
It usually sleeps alone but has been observed to sleep with several conspecifics (individuals of the same species), including other adults.
Despite its slow metabolism rate, the Sunda slow loris has a high-energy diet. Its slow lifestyle may be due to the energy costs of detoxifying certain secondary plant compounds in many genera of food plants their diets.
All slow loris species produce a toxin in glands on the insides of their elbows. This is spread across their bodies and those of their offspring using the toothcomb while grooming. When threatened with predators, the Sunda slow loris can bite, roll into a ball exposing its toxic saliva-covered fur, or roll up and drop from the trees.
However, the primary method of predator avoidance is crypsis, whereby it hides. The Asiatic reticulated python, the changeable hawk-eagle and the Sumatran orangutan have been recorded as predators of the Sunda slow loris.
One study showed only 8% of the species' active time was spent near other individuals. Where home ranges overlap, spatial groups are formed. These groups consist of one male, one female and up to three younger individuals. The interactions between these individuals are largely friendly; they include allogrooming, following, pant-growling, and click-calling, although social behaviors only make up around 3% of the activity budget.
When it comes into contact with conspecifics from other home ranges there is usually no reaction as home ranges are not defended.
In captivity however, it can be aggressive with other individuals. Males have shown antagonistic behaviors such as attacks, pursuits, threats, assertion, fighting, and subordination. The fighting often results in serious injury. Despite this, they are known to be generally sociable in captivity, with allogrooming being the most common social behavior.
There are eight distinct call types made by Sunda slow loris adults, which can be divided into two categories, contact and contact-seeking calls such as whistles and short keckers (a social play and attention-seeking call), and aggressive and defensive calls such as long keckers, screams, snarls and grunts.
Because they rely on crypsis to avoid predators, they do not make alarm calls. Infants emits clicks and squeaks when disturbed. During estrus, females make whistle calls when in visual contact with a male. When exploring new environments and during handling, it makes ultrasonic vocalisations out of the human hearing range.
Olfactory communication is very important in this species. It has roles in alerting others of the identity, physical state and position of the individual. It is also important in reproduction.
Like other slow lorises, the Sunda slow loris has glands on its elbows that exude oils. The gland is licked to spread scent and is thought to have evolved for communication, but it is toxic to humans. If the person is allergic to the animal they can go into shock and could even die. It also has glands on its anus
and can signal conspecifics by perineal rubbing which deposits urine.
When it locates the scent of another slow loris it may rub its face on the substrate where the scent was found. The Sunda slow loris may grin or bare its teeth. When stressed, infants may grin, while adults bear their teeth to show aggression or fear, but also during play.
Reproduction is one of the few times the Sunda slow loris aggregates with conspecifics, as it is largely solitary. One study recorded that the most slow lorises ever seen together was six, this appeared to be a female in estrus and five males following her.
This may suggest a more promiscuous mating system, where females mate with more than one male. Despite this, its testis size is small compared with similar sized promsimians, which is indicative of monogamy. In the wild the mating system of the Sunda slow loris is thought to vary between populations.
Sexual maturity is reached between the ages of 18 and 24 months in females, and can be reached by 17 months in males. It is polyestrous, having many periods of sexual receptivity during a year.
In captivity however, there is a clear birth peak between March and May. It has been observed that reproductive patterns of captive prosimians in the northern hemisphere are altered. The estrus cycle lasts 29–45 days, with most copulations occurring on the same day.
Females in estrus are followed by males, with copulation initiated by the female. The female will hang from a branch and may vocalise. The male will hold the female and the branch and copulate with her. Urine-marking and vocalising are also used by the female to solicit mating. The male may create a mating plug following copulation.
The gestation period averages 192.2 days, after which one young is born, although twinning has been observed. Both males and females will disperse in the wild, this occurs between 16 and 27 months.
The Sunda slow loris is found in continuous canopy tropical rainforests. It is adaptable and will also live in other types of habitat. It is found in Indonesia, on the islands of Sumatra, Batam and Galang in the Riau Archipelago, and Tebing Tinggi Island and Great Natuna (Bunguran) in the Natuna Islands; in Malaysia on the Malay Peninsula and Pulau Tioman; in the southern peninsular of Thailand; and Singapore.
The Sunda slow loris is sympatric shares its range with the Bengal slow loris in Thailand and
hybridisation has occurred.
Phylum: Chordata
Class: Mammalia
Order: Primates
Suborder: Strepsirrhini
Family: Lorisidae
Genus: Nycticebus
Species: N. coucang
Binomial name Nycticebus coucang
(Boddaert, 1785)
懒猴(学名 Nycticebus coucang)也叫做蜂猴,是懒猴属的一种。夜间活动,动作缓慢,栖息在树的顶部。食鸟、昆虫和野果等。
体型比家猫略小,尾极短,隐于毛丛中,不易看见,头圆耳小,眼大而圆,善于夜间看物体。体背和侧面毛呈棕褐色,背中央有一深栗红色纵纹;腹面灰白;四肢粗而短,指、趾具有指甲,仅后肢第二趾具爪,前肢第二指退化,仅剩一短指。
懒猴是目前已知唯一一种有毒的灵长类动物,在受到威胁时腋下会分泌一种呈棕色的油状物,可与唾液混合成有毒的物质(油状物和唾液本身也有毒性)。毒素通过食用有毒的昆虫,如蜈蚣蝎子等获得,所以只有野生的懒猴才可以分泌正常水平的毒性物质,而在保护区内养殖的懒猴因提供的食物一般不含毒素因此不能获取毒素和合成有毒物质。
主要分布于热带、亚热带森林中,共有三个已知亚种:
• 大懒猴 Nycticebus coucang coucang
• 婆罗洲懒猴 Nycticebus coucang menagensis
• 爪哇懒猴 Nycticebus coucang javanicus
体型比家猫略小,尾极短,隐于毛丛中,不易看见,头圆耳小,眼大而圆,善于夜间看物体。体背和侧面毛呈棕褐色,背中央有一深栗红色纵纹;腹面灰白;四肢粗而短,指、趾具有指甲,仅后肢第二趾具爪,前肢第二指退化,仅剩一短指。
懒猴是目前已知唯一一种有毒的灵长类动物,在受到威胁时腋下会分泌一种呈棕色的油状物,可与唾液混合成有毒的物质(油状物和唾液本身也有毒性)。毒素通过食用有毒的昆虫,如蜈蚣蝎子等获得,所以只有野生的懒猴才可以分泌正常水平的毒性物质,而在保护区内养殖的懒猴因提供的食物一般不含毒素因此不能获取毒素和合成有毒物质。
主要分布于热带、亚热带森林中,共有三个已知亚种:
• 大懒猴 Nycticebus coucang coucang
• 婆罗洲懒猴 Nycticebus coucang menagensis
• 爪哇懒猴 Nycticebus coucang javanicus
Iomys horsfieldii davisoni
Horsfield's flying squirrel
Horsfield's flying squirrel
Iomys is a small genus of rodent in the family Sciuridae.
Its two species are:
Javanese flying squirrel (Iomys horsfieldii) and
Mentawi flying squirrel (Iomys sipora)
Horsfield's Flying Squirrel, or Javanese Flying Squirrel (Iomys horsfieldii ) , is a medium-sized flying squirrel
which occurs in a range of habitats including primary forest and poor secondary forest.
Flying squirrels (scientifically known as Pteromyini or Petauristini) are a tribe of 50 species of squirrels in the family Sciuridae.
Despite their name, they are not in fact capable of full flight in the same way as birds or bats, but they are able to glide from one tree to another with the aid of a patagium, a furred parachute-like skin membrane that stretches from wrist to ankle.
Their long tails also provide stability as they glide. Anatomically they are very similar to other squirrels with a number of adaptations to suit their lifestyle; their limb bones are longer and their hand bones, foot bones, and distal vertebrae are shorter. Flying squirrels are able to steer and exert control over their glide path with their limbs and tail.
Molecular studies have shown that flying squirrels are monophyletic and originated some 18–20 million years ago. The genus Paracitellus is the earliest lineage to the flying squirrel dating back to the late Oligocene era.
Flying squirrels can easily forage for food in the night, given their highly developed sense of smell.
Most are nocturnal and omnivorous, eating fruit, seeds, buds, flowers, insects, gastropods, spiders, fungi, bird's eggs and tree sap. They eat according to their environment and will eat whatever food they can find.
The young are born in a nest and are at first naked and helpless. They are cared for by their mother and by five weeks are able to practice gliding skills so that by ten weeks they are ready to leave the nest.
The direction and speed of the animal in midair are varied by changing the positions of its limbs, largely controlled by small cartilaginous wrist bones.
There is a cartilage projection from the wrist that squirrel holds upwards during a glide.
This specialized cartilage is only present in flying squirrels and not other gliding mammals.
The life expectancy of flying squirrels in the wild is about six years, and flying squirrels can live up to fifteen years in zoos.
The mortality rate in young flying squirrels is high because of predators and diseases. Predators of flying squirrels include tree snakes, raccoons, owls, martens, fishers, coyotes, bobcats, and feral cats.
The mating season for flying squirrels is during February and March. When the infants are born, the female squirrels live with them in maternal nest sites. The mothers nurture and protect them until they leave the nest. The males do not participate in nurturing their offspring.
Many gliders have specialized diets and there is evidence to believe that gliders may be able to take advantage of scattered protein deficient food. Additionally, gliding is a fast form of locomotion and by reducing travel time between patches, they can increase the amount of foraging time.
I. horsfieldii are fairly adaptable animals. They are found in a wide variety of habitats. They can live in a variety of forest environments, even those that are degraded.
They also inhabit scrub and are found on durian plantations. They shelter in hollow trees and are limited to southern Asia. Their native range stretches from the peninsula of Malaysia down through Sumatra, Java, and Borneo. There have been additional sightings of individuals in Singapore, Tioman, and Penang.
Javanese flying squirrels are mostly orange-brown to brown-gray, with a tail that is a slightly brighter shade on the ventral surface and sides. Body length averages 182 mm and the tail is as long as, or slightly longer than, the body.
Like other flying squirrels, Javanese flying squirrels have a thin flap of skin, the patagium, connecting the fore and hind limbs to allow for gliding. The patagium is slightly darker in color than the rest of the body. Javanese flying squirrels also have relatively large eyes and ears and long claws for gripping the trees that they live in.
There are three named subspecies of Javanese flying squirrels, though too few specimens have been found to be certain whether these constitute distinct groups.
Iomys horsfieldii davisoni has dark brown-gray fur and a dark brown tail mottled with red-brown at the edges and a short tooth-row.
Iomys horsfieldii penangensis has lighter, brighter coloration and a longer tooth-row.
Iomys horsfieldii thomsoni is very similar to I. h. davisoni, but the tail is uniformly red-brown rather than mottled.
Javanese flying squirrels can be distinguished from their closest relatives by their large size, the orange tinge to the lighter underbelly, and the absence, or near absence of hair on the upper surface of the ear lobe.
The very similar Petinomys genibarbis can be distinguished from Javanese flying squirrels by the presence of a 5 mm wart on the cheek, sprouting long black whiskers.
Javanese flying squirrels are nocturnal and arboreal. Like other flying squirrels, they are probably able to glide long distances between trees and use their long claws for grip. Almost nothing is known about their social behavior. Members of the related genus Petaurista have been recorded foraging alone or in small groups, so it is possible that Javanese flying squirrels also organize themselves in this manner.
Phylum: Chordata
Class: Mammalia
Order: Rodentia
Family: Sciuridae
Subfamily: Sciurinae
Tribe: Pteromyini
Genus: Iomys Thomas, 1908
Species : Iomys horsfieldii davisoni
Its two species are:
Javanese flying squirrel (Iomys horsfieldii) and
Mentawi flying squirrel (Iomys sipora)
Horsfield's Flying Squirrel, or Javanese Flying Squirrel (Iomys horsfieldii ) , is a medium-sized flying squirrel
which occurs in a range of habitats including primary forest and poor secondary forest.
Flying squirrels (scientifically known as Pteromyini or Petauristini) are a tribe of 50 species of squirrels in the family Sciuridae.
Despite their name, they are not in fact capable of full flight in the same way as birds or bats, but they are able to glide from one tree to another with the aid of a patagium, a furred parachute-like skin membrane that stretches from wrist to ankle.
Their long tails also provide stability as they glide. Anatomically they are very similar to other squirrels with a number of adaptations to suit their lifestyle; their limb bones are longer and their hand bones, foot bones, and distal vertebrae are shorter. Flying squirrels are able to steer and exert control over their glide path with their limbs and tail.
Molecular studies have shown that flying squirrels are monophyletic and originated some 18–20 million years ago. The genus Paracitellus is the earliest lineage to the flying squirrel dating back to the late Oligocene era.
Flying squirrels can easily forage for food in the night, given their highly developed sense of smell.
Most are nocturnal and omnivorous, eating fruit, seeds, buds, flowers, insects, gastropods, spiders, fungi, bird's eggs and tree sap. They eat according to their environment and will eat whatever food they can find.
The young are born in a nest and are at first naked and helpless. They are cared for by their mother and by five weeks are able to practice gliding skills so that by ten weeks they are ready to leave the nest.
The direction and speed of the animal in midair are varied by changing the positions of its limbs, largely controlled by small cartilaginous wrist bones.
There is a cartilage projection from the wrist that squirrel holds upwards during a glide.
This specialized cartilage is only present in flying squirrels and not other gliding mammals.
The life expectancy of flying squirrels in the wild is about six years, and flying squirrels can live up to fifteen years in zoos.
The mortality rate in young flying squirrels is high because of predators and diseases. Predators of flying squirrels include tree snakes, raccoons, owls, martens, fishers, coyotes, bobcats, and feral cats.
The mating season for flying squirrels is during February and March. When the infants are born, the female squirrels live with them in maternal nest sites. The mothers nurture and protect them until they leave the nest. The males do not participate in nurturing their offspring.
Many gliders have specialized diets and there is evidence to believe that gliders may be able to take advantage of scattered protein deficient food. Additionally, gliding is a fast form of locomotion and by reducing travel time between patches, they can increase the amount of foraging time.
I. horsfieldii are fairly adaptable animals. They are found in a wide variety of habitats. They can live in a variety of forest environments, even those that are degraded.
They also inhabit scrub and are found on durian plantations. They shelter in hollow trees and are limited to southern Asia. Their native range stretches from the peninsula of Malaysia down through Sumatra, Java, and Borneo. There have been additional sightings of individuals in Singapore, Tioman, and Penang.
Javanese flying squirrels are mostly orange-brown to brown-gray, with a tail that is a slightly brighter shade on the ventral surface and sides. Body length averages 182 mm and the tail is as long as, or slightly longer than, the body.
Like other flying squirrels, Javanese flying squirrels have a thin flap of skin, the patagium, connecting the fore and hind limbs to allow for gliding. The patagium is slightly darker in color than the rest of the body. Javanese flying squirrels also have relatively large eyes and ears and long claws for gripping the trees that they live in.
There are three named subspecies of Javanese flying squirrels, though too few specimens have been found to be certain whether these constitute distinct groups.
Iomys horsfieldii davisoni has dark brown-gray fur and a dark brown tail mottled with red-brown at the edges and a short tooth-row.
Iomys horsfieldii penangensis has lighter, brighter coloration and a longer tooth-row.
Iomys horsfieldii thomsoni is very similar to I. h. davisoni, but the tail is uniformly red-brown rather than mottled.
Javanese flying squirrels can be distinguished from their closest relatives by their large size, the orange tinge to the lighter underbelly, and the absence, or near absence of hair on the upper surface of the ear lobe.
The very similar Petinomys genibarbis can be distinguished from Javanese flying squirrels by the presence of a 5 mm wart on the cheek, sprouting long black whiskers.
Javanese flying squirrels are nocturnal and arboreal. Like other flying squirrels, they are probably able to glide long distances between trees and use their long claws for grip. Almost nothing is known about their social behavior. Members of the related genus Petaurista have been recorded foraging alone or in small groups, so it is possible that Javanese flying squirrels also organize themselves in this manner.
Phylum: Chordata
Class: Mammalia
Order: Rodentia
Family: Sciuridae
Subfamily: Sciurinae
Tribe: Pteromyini
Genus: Iomys Thomas, 1908
Species : Iomys horsfieldii davisoni
Manis javanica
The Sunda pangolin
The Sunda pangolin
Manidae is the only extant family of pangolins from superfamily Manoidea. This family comprises three genera (Manis from subfamily Maninae, Phataginus from subfamily Phatagininae, and Smutsia from subfamily Smutsiinae), as well as extinct Fayum pangolin.
All species of living pangolin had been assigned to the genus Manis until the late 2000s, when research prompted the splitting of extant pangolins into three genera: Manis, Phataginus, and Smutsia.
The name Sunda is a reference to the pangolin’s range throughout a biogeographical region called Sundaland that comprises the Malay Peninsula and the Indonesian Archipelago islands west of the Wallace Line. Pangolins are specialist myrmecophages.
Myrmecophagy is a feeding behavior defined by the consumption of termites or ants, particularly as pertaining to those animal species whose diets are largely or exclusively composed of said insect types.
Literally, myrmecophagy means "ant eating" (Ancient Greek: murmēx, "ants" and phagein, "to eat") rather than "termite eating" (for which the strict term is termitophagy). The two habits often overlap, as both of these eusocial insect types often live in large, densely populated nests requiring similar adaptations in the animal species that exploit them.
M.javanica using strong claws on their forelimbs to break into nests and mounds found underground or in trees. Once found, they use their long sticky tongue to gather the insects.
The Sunda pangolin (Manis javanica), also known as the Malayan or Javan pangolin, is a species of pangolin.
It is found throughout Southeast Asia, including Myanmar, Thailand, Cambodia, Laos, Malaysia, Singapore, Vietnam, and the islands of Borneo, Java, Sumatra and the Lesser Sunda Islands. It prefers forested habitats (primary, secondary, and scrub forest) and plantations (rubber, palm oil). A large part of its life is spent in trees.
In the past, this species has included the closely related Palawan pangolin (M. culionensis), as both are in the subgenus Paramanis. It is closely related to the Chinese pangolin, although the Malayan species is larger, lighter in colour, and has shorter fore claws.
The skin of the Sunda pangolin's feet is granular, although pads are found on its front feet. It has thick and powerful claws to dig into the soils in search of ant nests or to tear into termite mounds.
The Sunda pangolin has poor eyesight, but a highly developed sense of smell. Lacking teeth, its long, sticky tongue serves to collect ants and termites. Its body is covered by rows of scales and fibrous hair.
The head-body length of this pangolin can measure 40–65 cm, tail length is 35–56 cm, and its weight is up to 10 kg. Males are larger than females.
Pangolins are solitary mammals and are not dangerous or aggressive. Looks can be deceiving as these hard-scaled ‘ant-eaters’ are actually quite placid and go about their business without much fuss and usually in complete silence.
Pangolins are also one of the few animals that have no vocal cords, so the only sounds they produce are from huffing and puffing their way through a tasty meal.
Pangolins make very few sounds. When they walk or climb, their scales can be heard rubbing against one another and against the vegetation. When awakening or feeding, they snort and chuff audibly. Males also rarely make a soft hooting noise.
Pangolins give birth annually to one or two offspring. They breed in the autumn, and females give birth in the winter burrow. Den preference has been known to shift at this time to favor mature forest tree hollows. Presumably, these hollows offer more fortification and stability for the decreased mobility that comes with birthing and caring for young.
The amount of time the mother will spend at any one of these dens will increase during parental care periods. Parental care is given for about three months. In these three months, the range of the mother Pangolin drastically decreases as she travels and forages with her offspring. Only a few weeks before the offspring becomes fully independent, the mother and her young can display brief spikes in diurnal activity.
Pangolins are sometimes found in pairs, but normally they are solitary, nocturnal, and behave timidly. They protect their soft underparts by rolling into balls when they feel threatened. They are strong diggers and make burrows lined with vegetation for insulation near termite mounds and ant nests.
Sunda pangolins have low immunity, making them sensitive to fluctuations in temperature.
The Sunda pangolin's main predators are humans, tigers, and the clouded leopard.
Pangolins as a family are among the most heavily poached and exploited protected animals. Like other pangolin species, the Sunda pangolin is hunted for its skin, scales, and meat, used in clothing manufacture and traditional medicine.
Scales are made into rings as charms against rheumatic fever, and meat is eaten by indigenous peoples. Despite enjoying protected status almost everywhere in its range, illegal international trade, largely driven by Chinese buyers, has led to rapidly decreasing population numbers. The Sunda pangolin is currently considered to be critically endangered.
As of 2016, all pangolin species are listed on CITES Appendix I, which prohibits commercial international trade of wild-caught specimens or their body parts. China raised the protection status of all pangolin species to the highest level in 2020.
A metagenomic study published in 2019 previously revealed that SARS-CoV, the strain of the virus that causes SARS, was the most widely distributed coronavirus among a sample of Sunda pangolins.
On 7 February 2020, it was announced that researchers from Guangzhou had discovered a pangolin sample with a viral nucleic acid sequence "99% identical" to SARS-CoV-2. When released, the results clarified that "the receptor-binding domain of the S protein of the newly discovered Pangolin-CoV is virtually identical to that of 2019-nCoV, with one amino acid difference." Pangolins are protected under Chinese law, but their poaching and trading for use in traditional Chinese medicine remains common.
Pangolin coronaviruses found to date only share at most 92% of their whole genomes with SARS-CoV-2, making them less similar than RaTG13 to SARS-CoV-2. This is insufficient to prove pangolins to be the intermediate host; in comparison, the SARS virus responsible for the 2002–2004 outbreak shared 99.8% of its genome with a known civet coronavirus.
Phylum: Chordata
Class: Mammalia
Order: Pholidota
Family: Manidae
Genus: Manis
Subgenus: Paramanis
Species: M. javanica
Binomial name Manis javanica
Desmarest, 1822
All species of living pangolin had been assigned to the genus Manis until the late 2000s, when research prompted the splitting of extant pangolins into three genera: Manis, Phataginus, and Smutsia.
The name Sunda is a reference to the pangolin’s range throughout a biogeographical region called Sundaland that comprises the Malay Peninsula and the Indonesian Archipelago islands west of the Wallace Line. Pangolins are specialist myrmecophages.
Myrmecophagy is a feeding behavior defined by the consumption of termites or ants, particularly as pertaining to those animal species whose diets are largely or exclusively composed of said insect types.
Literally, myrmecophagy means "ant eating" (Ancient Greek: murmēx, "ants" and phagein, "to eat") rather than "termite eating" (for which the strict term is termitophagy). The two habits often overlap, as both of these eusocial insect types often live in large, densely populated nests requiring similar adaptations in the animal species that exploit them.
M.javanica using strong claws on their forelimbs to break into nests and mounds found underground or in trees. Once found, they use their long sticky tongue to gather the insects.
The Sunda pangolin (Manis javanica), also known as the Malayan or Javan pangolin, is a species of pangolin.
It is found throughout Southeast Asia, including Myanmar, Thailand, Cambodia, Laos, Malaysia, Singapore, Vietnam, and the islands of Borneo, Java, Sumatra and the Lesser Sunda Islands. It prefers forested habitats (primary, secondary, and scrub forest) and plantations (rubber, palm oil). A large part of its life is spent in trees.
In the past, this species has included the closely related Palawan pangolin (M. culionensis), as both are in the subgenus Paramanis. It is closely related to the Chinese pangolin, although the Malayan species is larger, lighter in colour, and has shorter fore claws.
The skin of the Sunda pangolin's feet is granular, although pads are found on its front feet. It has thick and powerful claws to dig into the soils in search of ant nests or to tear into termite mounds.
The Sunda pangolin has poor eyesight, but a highly developed sense of smell. Lacking teeth, its long, sticky tongue serves to collect ants and termites. Its body is covered by rows of scales and fibrous hair.
The head-body length of this pangolin can measure 40–65 cm, tail length is 35–56 cm, and its weight is up to 10 kg. Males are larger than females.
Pangolins are solitary mammals and are not dangerous or aggressive. Looks can be deceiving as these hard-scaled ‘ant-eaters’ are actually quite placid and go about their business without much fuss and usually in complete silence.
Pangolins are also one of the few animals that have no vocal cords, so the only sounds they produce are from huffing and puffing their way through a tasty meal.
Pangolins make very few sounds. When they walk or climb, their scales can be heard rubbing against one another and against the vegetation. When awakening or feeding, they snort and chuff audibly. Males also rarely make a soft hooting noise.
Pangolins give birth annually to one or two offspring. They breed in the autumn, and females give birth in the winter burrow. Den preference has been known to shift at this time to favor mature forest tree hollows. Presumably, these hollows offer more fortification and stability for the decreased mobility that comes with birthing and caring for young.
The amount of time the mother will spend at any one of these dens will increase during parental care periods. Parental care is given for about three months. In these three months, the range of the mother Pangolin drastically decreases as she travels and forages with her offspring. Only a few weeks before the offspring becomes fully independent, the mother and her young can display brief spikes in diurnal activity.
Pangolins are sometimes found in pairs, but normally they are solitary, nocturnal, and behave timidly. They protect their soft underparts by rolling into balls when they feel threatened. They are strong diggers and make burrows lined with vegetation for insulation near termite mounds and ant nests.
Sunda pangolins have low immunity, making them sensitive to fluctuations in temperature.
The Sunda pangolin's main predators are humans, tigers, and the clouded leopard.
Pangolins as a family are among the most heavily poached and exploited protected animals. Like other pangolin species, the Sunda pangolin is hunted for its skin, scales, and meat, used in clothing manufacture and traditional medicine.
Scales are made into rings as charms against rheumatic fever, and meat is eaten by indigenous peoples. Despite enjoying protected status almost everywhere in its range, illegal international trade, largely driven by Chinese buyers, has led to rapidly decreasing population numbers. The Sunda pangolin is currently considered to be critically endangered.
As of 2016, all pangolin species are listed on CITES Appendix I, which prohibits commercial international trade of wild-caught specimens or their body parts. China raised the protection status of all pangolin species to the highest level in 2020.
A metagenomic study published in 2019 previously revealed that SARS-CoV, the strain of the virus that causes SARS, was the most widely distributed coronavirus among a sample of Sunda pangolins.
On 7 February 2020, it was announced that researchers from Guangzhou had discovered a pangolin sample with a viral nucleic acid sequence "99% identical" to SARS-CoV-2. When released, the results clarified that "the receptor-binding domain of the S protein of the newly discovered Pangolin-CoV is virtually identical to that of 2019-nCoV, with one amino acid difference." Pangolins are protected under Chinese law, but their poaching and trading for use in traditional Chinese medicine remains common.
Pangolin coronaviruses found to date only share at most 92% of their whole genomes with SARS-CoV-2, making them less similar than RaTG13 to SARS-CoV-2. This is insufficient to prove pangolins to be the intermediate host; in comparison, the SARS virus responsible for the 2002–2004 outbreak shared 99.8% of its genome with a known civet coronavirus.
Phylum: Chordata
Class: Mammalia
Order: Pholidota
Family: Manidae
Genus: Manis
Subgenus: Paramanis
Species: M. javanica
Binomial name Manis javanica
Desmarest, 1822
Asian palm civet 亚洲棕榈果子狸
(Paradoxurus hermaphroditus)
(Paradoxurus hermaphroditus)
The Asian palm civet (Paradoxurus hermaphroditus), also called common palm civet, toddy cat and musang, is a viverrid native to South and Southeast Asia.
Since 2008, it is IUCN Red Listed as Least Concern as it accommodates to a broad range of habitats. It is widely distributed with large populations that in 2008 were thought unlikely to be declining.
In Indonesia, it is threatened by poaching and illegal wildlife trade; buyers use it for the increasing production of kopi luwak.
The Asian palm civet's long, stocky body is covered with coarse, shaggy hair that is usually greyish in colour. It has a white mask across the forehead, a small white patch under each eye, a white spot on each side of the nostrils, and a narrow dark line between the eyes. The muzzle, ears, lower legs, and distal half of the tail are black, with three rows of black markings on the body. Its head-to-body length is about 53 cm (21 in) with a 48 cm (19 in) long unringed tail. It weighs 2 to 5 kg (4 to 11 lb). Its anal scent glands emit a nauseating secretion as a chemical defense when threatened or upset.
Asian palm civet is native to India, Nepal, Bangladesh,
Bhutan, Myanmar, Sri Lanka, Thailand, Singapore, Peninsular Malaysia, Sabah, Sarawak, Laos, Cambodia,
Brunei Darussalam, Vietnam, China, the Philippines, and the Indonesian islands of Sumatra, Java, Bawean,
Kalimantan and Siberut. It was introduced to Irian Jaya, the Lesser Sunda Islands, Maluku, and Sulawesi.
It usually inhabits primary forests, but also occurs at lower densities in secondary and selectively logged forest. It is also present in parks and suburban gardens with mature fruit trees, fig trees, and undisturbed vegetation. Its sharp claws allow climbing of trees and house gutters.
In most parts of Sri Lanka, palm civets are considered a nuisance since they litter in ceilings and attics of common households, and make loud noises fighting and moving about at night.
The Asian palm civet is thought to lead a solitary lifestyle, except for brief periods during mating. It is both terrestrial and arboreal, showing a nocturnal activity pattern with peaks between late evening until after midnight. It is usually active between dawn and 4:00 in the morning, but less active during nights when the moon is brightest.
Scent marking behaviour and olfactory response to various excretions such as urine, feces, and secretion of the perineal gland differs in males and females. Scent marking by dragging the perineal gland and leaving the secretion on the substrate was most commonly observed in animals of both sexes.
The duration of the olfactory response varied and depended both on the sex and excretion type. The palm civet can distinguish animal species, sex, familiar and unfamiliar individuals by the odor of the perineal gland secretion.
The Asian palm civet is an omnivore feeding foremost on fruits such as berries and pulpy fruits. It thus helps to maintain tropical forest ecosystems via seed dispersal.
It eats chiku, mango, rambutan, and coffee, but also small mammals and insects. It plays an important role in the natural regeneration of Pinanga kuhlii and P. zavana palms at Gunung Gede Pangrango National Park. It also feeds on palm flower sap, which when fermented becomes palm wine, a sweet liquor ("toddy"). Because of this habit, it is called the toddy cat. Due to its solitary and nocturnal habits, little is known about its reproductive processes and behaviour.
In some parts of its range Asian palm civets are hunted for bushmeat and the pet trade. In southern China it is extensively hunted and trapped. Dead individuals were found with local tribes where it is killed for its meat, in Coimbatore, Tamil Nadu, and Agra, Uttar Pradesh, between 1998 and 2003 in India.
The oil extracted from small pieces of the meat kept in linseed oil in a closed earthen pot and regularly sunned, is used indigenously as a cure for scabies.
Scabies is an infestation of the skin by the human itch mite (Sarcoptes scabiei var. hominis). The microscopic scabies mite burrows into the upper layer of the skin where it lives and lays its eggs. The most common symptoms of scabies are intense itching and a pimple-like skin rash. Symptoms: Itch; Rash
Kopi luwak is coffee prepared using coffee beans that have been subjected to ingestion and fermentation in the gastrointestinal tract of the Asian palm civet, which is called luwak in Indonesia. Caffeine content in both Arabica and Robusta luwak coffee is lower than in unfermented coffee. Large deformation mechanical rheology testing revealed that civet coffee beans are harder and more brittle in nature than their control counterparts indicating that digestive juices enter into the beans and modify the micro-structural properties of these beans. Proteolytic enzymes cause substantial breakdown of storage proteins.
Kopi luwak is traditionally made from the faeces of wild civets, however, due to it becoming a trendy drink, civets are being increasingly captured from the wild and fed coffee beans to mass-produce this blend. Many of these civets are housed in battery cage systems which have been criticised on animal welfare grounds. The impact of the demand for this fashionable coffee on wild palm civet populations is yet unknown but may constitute a significant threat. In Indonesia, the demand for Asian palm civets appears to be in violation of the quota set for pets.
The Asian palm civet (Paradoxurus hermaphroditus) is listed on CITES Appendix III. There is a quota in place in Indonesia, precluding trade from certain areas, setting a cap on the number of civets that can be taken from the wild, and allowing only 10% of those removed from the wild to be sold domestically. This quota is largely ignored by hunters and traders and is not enforced by authorities.
This species has become popular as a pet in Indonesia in recent years, causing a rise in the numbers found in markets in Java and Bali. The majority of the animals sold as pets originate from the wild. The high numbers of animals seen, lack of adherence to the quota and lack of enforcement of the laws are causes for conservation concern.
Phylum: Chordata
Class: Mammalia
Order: Carnivora
Suborder: Feliformia
Family: Viverridae
Genus: Paradoxurus
Species: P. hermaphroditus
Binomial name Paradoxurus hermaphroditus
(Pallas, 1777)
Since 2008, it is IUCN Red Listed as Least Concern as it accommodates to a broad range of habitats. It is widely distributed with large populations that in 2008 were thought unlikely to be declining.
In Indonesia, it is threatened by poaching and illegal wildlife trade; buyers use it for the increasing production of kopi luwak.
The Asian palm civet's long, stocky body is covered with coarse, shaggy hair that is usually greyish in colour. It has a white mask across the forehead, a small white patch under each eye, a white spot on each side of the nostrils, and a narrow dark line between the eyes. The muzzle, ears, lower legs, and distal half of the tail are black, with three rows of black markings on the body. Its head-to-body length is about 53 cm (21 in) with a 48 cm (19 in) long unringed tail. It weighs 2 to 5 kg (4 to 11 lb). Its anal scent glands emit a nauseating secretion as a chemical defense when threatened or upset.
Asian palm civet is native to India, Nepal, Bangladesh,
Bhutan, Myanmar, Sri Lanka, Thailand, Singapore, Peninsular Malaysia, Sabah, Sarawak, Laos, Cambodia,
Brunei Darussalam, Vietnam, China, the Philippines, and the Indonesian islands of Sumatra, Java, Bawean,
Kalimantan and Siberut. It was introduced to Irian Jaya, the Lesser Sunda Islands, Maluku, and Sulawesi.
It usually inhabits primary forests, but also occurs at lower densities in secondary and selectively logged forest. It is also present in parks and suburban gardens with mature fruit trees, fig trees, and undisturbed vegetation. Its sharp claws allow climbing of trees and house gutters.
In most parts of Sri Lanka, palm civets are considered a nuisance since they litter in ceilings and attics of common households, and make loud noises fighting and moving about at night.
The Asian palm civet is thought to lead a solitary lifestyle, except for brief periods during mating. It is both terrestrial and arboreal, showing a nocturnal activity pattern with peaks between late evening until after midnight. It is usually active between dawn and 4:00 in the morning, but less active during nights when the moon is brightest.
Scent marking behaviour and olfactory response to various excretions such as urine, feces, and secretion of the perineal gland differs in males and females. Scent marking by dragging the perineal gland and leaving the secretion on the substrate was most commonly observed in animals of both sexes.
The duration of the olfactory response varied and depended both on the sex and excretion type. The palm civet can distinguish animal species, sex, familiar and unfamiliar individuals by the odor of the perineal gland secretion.
The Asian palm civet is an omnivore feeding foremost on fruits such as berries and pulpy fruits. It thus helps to maintain tropical forest ecosystems via seed dispersal.
It eats chiku, mango, rambutan, and coffee, but also small mammals and insects. It plays an important role in the natural regeneration of Pinanga kuhlii and P. zavana palms at Gunung Gede Pangrango National Park. It also feeds on palm flower sap, which when fermented becomes palm wine, a sweet liquor ("toddy"). Because of this habit, it is called the toddy cat. Due to its solitary and nocturnal habits, little is known about its reproductive processes and behaviour.
In some parts of its range Asian palm civets are hunted for bushmeat and the pet trade. In southern China it is extensively hunted and trapped. Dead individuals were found with local tribes where it is killed for its meat, in Coimbatore, Tamil Nadu, and Agra, Uttar Pradesh, between 1998 and 2003 in India.
The oil extracted from small pieces of the meat kept in linseed oil in a closed earthen pot and regularly sunned, is used indigenously as a cure for scabies.
Scabies is an infestation of the skin by the human itch mite (Sarcoptes scabiei var. hominis). The microscopic scabies mite burrows into the upper layer of the skin where it lives and lays its eggs. The most common symptoms of scabies are intense itching and a pimple-like skin rash. Symptoms: Itch; Rash
Kopi luwak is coffee prepared using coffee beans that have been subjected to ingestion and fermentation in the gastrointestinal tract of the Asian palm civet, which is called luwak in Indonesia. Caffeine content in both Arabica and Robusta luwak coffee is lower than in unfermented coffee. Large deformation mechanical rheology testing revealed that civet coffee beans are harder and more brittle in nature than their control counterparts indicating that digestive juices enter into the beans and modify the micro-structural properties of these beans. Proteolytic enzymes cause substantial breakdown of storage proteins.
Kopi luwak is traditionally made from the faeces of wild civets, however, due to it becoming a trendy drink, civets are being increasingly captured from the wild and fed coffee beans to mass-produce this blend. Many of these civets are housed in battery cage systems which have been criticised on animal welfare grounds. The impact of the demand for this fashionable coffee on wild palm civet populations is yet unknown but may constitute a significant threat. In Indonesia, the demand for Asian palm civets appears to be in violation of the quota set for pets.
The Asian palm civet (Paradoxurus hermaphroditus) is listed on CITES Appendix III. There is a quota in place in Indonesia, precluding trade from certain areas, setting a cap on the number of civets that can be taken from the wild, and allowing only 10% of those removed from the wild to be sold domestically. This quota is largely ignored by hunters and traders and is not enforced by authorities.
This species has become popular as a pet in Indonesia in recent years, causing a rise in the numbers found in markets in Java and Bali. The majority of the animals sold as pets originate from the wild. The high numbers of animals seen, lack of adherence to the quota and lack of enforcement of the laws are causes for conservation concern.
Phylum: Chordata
Class: Mammalia
Order: Carnivora
Suborder: Feliformia
Family: Viverridae
Genus: Paradoxurus
Species: P. hermaphroditus
Binomial name Paradoxurus hermaphroditus
(Pallas, 1777)
Sambar deer 水鹿
(Rusa unicolor)
(Rusa unicolor)
The sambar (Rusa unicolor) is a large deer native to the Indian subcontinent, South China, and Southeast Asia that is listed as a vulnerable species on the IUCN Red List since 2008. Populations have declined substantially due to severe hunting, local insurgency, and industrial exploitation of habitat.
The appearance and the size of the sambar vary widely across its range, which has led to considerable taxonomic confusion in the past; over 40 different scientific synonyms have been used for the species.
In general, they attain a height of 102 to 160 cm (40 to 63 in) at the shoulder and may weigh as much as 546 kg (1,204 lb), though more typically 100 to 350 kg (220 to 770 lb).
Head and body length varies from 1.62 to 2.7 m (5.3 to 8.9 ft), with a 22 to 35 cm (8.7 to 13.8 in) tail.
Individuals belonging to western subspecies tend to be larger than those from the east, and females are smaller than males. Among all living cervid species, only the moose and the elk can attain larger sizes.
The large, rugged antlers are typically rusine, the brow tines being simple and the beams forked at the tip, so they have only three tines. The antlers are typically up to 110 cm (43 in) long in fully adult individuals. As with most deer, only the males have antlers. Males have 40 inches long antlers that are divided in three branches.
The shaggy coat can be from yellowish brown to dark grey in colour, and while it is usually uniform in colour, some subspecies have chestnut marks on the rump and underparts. Sambar also have a small but dense mane, which tends to be more prominent in males. The tail is relatively long for deer, and is generally black above with a whitish underside.
Adult males and pregnant or lactating females possess an unusual hairless, blood-red spot located about halfway down the underside of their throats. This sometimes oozes a white liquid, and is apparently glandular in nature.
The sambar is distributed in much of South Asia as far north as the south-facing slopes of the Himalayas in Nepal and India, in mainland Southeast Asia including Burma, Thailand, Indochina, the Malay Peninsula, Indonesia (Sumatra and Borneo), Taiwan, and South China, including Hainan.
It inhabits tropical dry forests, tropical seasonal forests, subtropical mixed forests with stands of conifers and montane grasslands, broadleaved deciduous and broadleaved evergreen trees, to tropical rainforests, and seldom moves far from water sources.
The sambar prefers the dense cover of deciduous shrubs and grasses, although the exact nature of this varies enormously with the environment because of its wide Asian range.
Adult male sambar can significantly damage plants, removing most branches on some shrubs and sometimes girdling trees by thrashing their antlers on shrubs and sapling trees. Its diet consist of bamboo shoots, grass, seedlings, fruits and plants. They leave territorial marks to advertise their territory.
Sambar deer have excellent senses of hearing and smell which are used mainly for detection of predators. Sambar deer are either crepuscular (active at dusk and dawn) or nocturnal (active during the night) animals.
Sambar deer are excellent swimmers and they are either solitary or live in groups composed of less than 10 members. Groups are usually composed of animals of one gender.
Sambar deer produce barking calls in the case of danger. Males also vocalize loudly to attract females during the mating season. Males occupy territory of around 1500 hectares, while females live on a territory of 300 hectares.
They use urine and scent to mark their territories.Sambar deer can mate throughout the whole year, but they prefer period from September to January. Males release smelly substance to attract females. They fight with each other before they get opportunity to mate. One male often mates with more than 6 females.
Pregnancy lasts 8 to 9 months and ends with one baby (rarely two). Baby is born without spots and marks on the body. They appear later in life. Young animal stays with its mother 2 years.bSambar deer can survive 20 years in the wild and up to 26 years in captivity.
Phylum: Chordata
Class: Mammalia
Order: Artiodactyla
Family: Cervidae
Subfamily: Cervinae
Genus: Rusa
Species: R. unicolor
Binomial name Rusa unicolor
The appearance and the size of the sambar vary widely across its range, which has led to considerable taxonomic confusion in the past; over 40 different scientific synonyms have been used for the species.
In general, they attain a height of 102 to 160 cm (40 to 63 in) at the shoulder and may weigh as much as 546 kg (1,204 lb), though more typically 100 to 350 kg (220 to 770 lb).
Head and body length varies from 1.62 to 2.7 m (5.3 to 8.9 ft), with a 22 to 35 cm (8.7 to 13.8 in) tail.
Individuals belonging to western subspecies tend to be larger than those from the east, and females are smaller than males. Among all living cervid species, only the moose and the elk can attain larger sizes.
The large, rugged antlers are typically rusine, the brow tines being simple and the beams forked at the tip, so they have only three tines. The antlers are typically up to 110 cm (43 in) long in fully adult individuals. As with most deer, only the males have antlers. Males have 40 inches long antlers that are divided in three branches.
The shaggy coat can be from yellowish brown to dark grey in colour, and while it is usually uniform in colour, some subspecies have chestnut marks on the rump and underparts. Sambar also have a small but dense mane, which tends to be more prominent in males. The tail is relatively long for deer, and is generally black above with a whitish underside.
Adult males and pregnant or lactating females possess an unusual hairless, blood-red spot located about halfway down the underside of their throats. This sometimes oozes a white liquid, and is apparently glandular in nature.
The sambar is distributed in much of South Asia as far north as the south-facing slopes of the Himalayas in Nepal and India, in mainland Southeast Asia including Burma, Thailand, Indochina, the Malay Peninsula, Indonesia (Sumatra and Borneo), Taiwan, and South China, including Hainan.
It inhabits tropical dry forests, tropical seasonal forests, subtropical mixed forests with stands of conifers and montane grasslands, broadleaved deciduous and broadleaved evergreen trees, to tropical rainforests, and seldom moves far from water sources.
The sambar prefers the dense cover of deciduous shrubs and grasses, although the exact nature of this varies enormously with the environment because of its wide Asian range.
Adult male sambar can significantly damage plants, removing most branches on some shrubs and sometimes girdling trees by thrashing their antlers on shrubs and sapling trees. Its diet consist of bamboo shoots, grass, seedlings, fruits and plants. They leave territorial marks to advertise their territory.
Sambar deer have excellent senses of hearing and smell which are used mainly for detection of predators. Sambar deer are either crepuscular (active at dusk and dawn) or nocturnal (active during the night) animals.
Sambar deer are excellent swimmers and they are either solitary or live in groups composed of less than 10 members. Groups are usually composed of animals of one gender.
Sambar deer produce barking calls in the case of danger. Males also vocalize loudly to attract females during the mating season. Males occupy territory of around 1500 hectares, while females live on a territory of 300 hectares.
They use urine and scent to mark their territories.Sambar deer can mate throughout the whole year, but they prefer period from September to January. Males release smelly substance to attract females. They fight with each other before they get opportunity to mate. One male often mates with more than 6 females.
Pregnancy lasts 8 to 9 months and ends with one baby (rarely two). Baby is born without spots and marks on the body. They appear later in life. Young animal stays with its mother 2 years.bSambar deer can survive 20 years in the wild and up to 26 years in captivity.
Phylum: Chordata
Class: Mammalia
Order: Artiodactyla
Family: Cervidae
Subfamily: Cervinae
Genus: Rusa
Species: R. unicolor
Binomial name Rusa unicolor
Usuage of a Deer Antlers
Antlers are bony skeletal protuberances of the skull, and consist mainly of the protein collagen and the mineral calcium hydroxyapatite.
Deer antlers are edible, and not just as a pill used in eastern medicine or a health supplement. Antlers can be used to make gelatin by boiling the ground antler and straining off the remains, which could be used to make fruit gelatin or added to homemade jellies. Processed antlers can also be used in baking recipes.
Sambar deer (R. u. unicolor) has the largest antlers of the species, which go through an annual cycle of growth, branching, hardening, and eventually falling off.
Each antler grows from an attachment point on the skull called a pedicle. While an antler is growing, it is covered with highly vascular skin called velvet, which supplies oxygen and nutrients to the growing bone.
Antlers are considered one of the most exaggerated cases of male secondary sexual traits in the animal kingdom, and grow faster than any other mammal bone.
Growth occurs at the tip, and is initially cartilage, which is later replaced by bone tissue. Once the antler has achieved its full size, the velvet is lost and the antler's bone dies. This dead bone structure is the mature antler.
In most cases, the bone at the base is destroyed by osteoclasts (Osteoclasts are the cells that degrade bone to initiate normal bone remodeling and mediate bone loss in pathologic conditions by increasing their resorptive activity. They are derived from precursors in the myeloid/monocyte lineage that circulate in the blood after their formation in the bone marrow ) and the antlers fall off at some point.
As a result of their fast growth rate, antlers are considered a handicap since there is an immense nutritional demand on deer to re-grow antlers annually, and thus can be honest signals of metabolic efficiency and food gathering capability.
Sambars in the ‘hard antler’ stage have the highest hormone concentrations and the sudden drop triggers antler casting.”
The stage when the antlers hardened coincide with the start of the period rutting or breeding season, when the males begin jousting with each other. (Female sambar, like nearly all other deer species, don’t grow antlers.) The hard antlers were recorded throughout the breeding season, lasting for about four to five months, generally from September to January.
In the velvet antler stage, antlers of elk and deer have been used in Asia as a dietary supplement or alternative medicinal substance for more than 2,000 years.
Deer antler extract has become popular among Western athletes and body builders because the extract, with its trace amounts of IGF-1, (Insulin-like growth factor 1 (IGF-1), also called somatomedin C, is a hormone similar in molecular structure to insulin which plays an important role in childhood growth, and has anabolic effects in adults) is believed to help build and repair muscle tissue; however, one double-blind study did not find evidence of intended effects in adults.
Traditional medical reports and clinical observations show that antler is biologically active to cure various diseases.
To make antler products acceptable as nutraceuticals and functional foods, chemical and biological properties of velvet antlers have to be clearly determined.
Antlers are made of chemical components consisting of sugars, fatty acids, amino acids, and nucleotides as essential molecules, which become macromolecules such as polysaccharides, lipids, proteins and nucleic acids, respectively.
For their physicochemical properties, each of these macromolecules is responsible for not only antler growth and development, but also biomedical and nutraceuticals uses of antlers.
Therefore, understanding chemical and molecular characteristics of antlers is crucially important to elucidate the clinical and medicinal efficacies of antlers. Hence, the review highlights information about various species of deer, its farming, antler preparation, antler composition, its traditional uses and scientific substantiation to it, dose and its future scope.
Courtesy source from:
Dr. Durgacharan Arun Bhagwat
Although rare now, some old recipes still call for Hartshorn. This product is made from distilled, ground antler by burning in kilns, which makes ammonium carbonate. This was the predecessor to baking powder, which is still used today in thin shaped cookies and crackers. When heated, the gaseous ammonia is released in the baking process, acting as a leveling agent.
Although you would not just bite into a shed antler or boil it up and put it on a plate, recently antlers have grown in popularity as dog chews. This is now a large part of the shed antler market. Dogs enjoy gnawing on them and get calcium as an added bonus.
Deer antlers are edible, and not just as a pill used in eastern medicine or a health supplement. Antlers can be used to make gelatin by boiling the ground antler and straining off the remains, which could be used to make fruit gelatin or added to homemade jellies. Processed antlers can also be used in baking recipes.
Sambar deer (R. u. unicolor) has the largest antlers of the species, which go through an annual cycle of growth, branching, hardening, and eventually falling off.
Each antler grows from an attachment point on the skull called a pedicle. While an antler is growing, it is covered with highly vascular skin called velvet, which supplies oxygen and nutrients to the growing bone.
Antlers are considered one of the most exaggerated cases of male secondary sexual traits in the animal kingdom, and grow faster than any other mammal bone.
Growth occurs at the tip, and is initially cartilage, which is later replaced by bone tissue. Once the antler has achieved its full size, the velvet is lost and the antler's bone dies. This dead bone structure is the mature antler.
In most cases, the bone at the base is destroyed by osteoclasts (Osteoclasts are the cells that degrade bone to initiate normal bone remodeling and mediate bone loss in pathologic conditions by increasing their resorptive activity. They are derived from precursors in the myeloid/monocyte lineage that circulate in the blood after their formation in the bone marrow ) and the antlers fall off at some point.
As a result of their fast growth rate, antlers are considered a handicap since there is an immense nutritional demand on deer to re-grow antlers annually, and thus can be honest signals of metabolic efficiency and food gathering capability.
Sambars in the ‘hard antler’ stage have the highest hormone concentrations and the sudden drop triggers antler casting.”
The stage when the antlers hardened coincide with the start of the period rutting or breeding season, when the males begin jousting with each other. (Female sambar, like nearly all other deer species, don’t grow antlers.) The hard antlers were recorded throughout the breeding season, lasting for about four to five months, generally from September to January.
In the velvet antler stage, antlers of elk and deer have been used in Asia as a dietary supplement or alternative medicinal substance for more than 2,000 years.
Deer antler extract has become popular among Western athletes and body builders because the extract, with its trace amounts of IGF-1, (Insulin-like growth factor 1 (IGF-1), also called somatomedin C, is a hormone similar in molecular structure to insulin which plays an important role in childhood growth, and has anabolic effects in adults) is believed to help build and repair muscle tissue; however, one double-blind study did not find evidence of intended effects in adults.
Traditional medical reports and clinical observations show that antler is biologically active to cure various diseases.
To make antler products acceptable as nutraceuticals and functional foods, chemical and biological properties of velvet antlers have to be clearly determined.
Antlers are made of chemical components consisting of sugars, fatty acids, amino acids, and nucleotides as essential molecules, which become macromolecules such as polysaccharides, lipids, proteins and nucleic acids, respectively.
For their physicochemical properties, each of these macromolecules is responsible for not only antler growth and development, but also biomedical and nutraceuticals uses of antlers.
Therefore, understanding chemical and molecular characteristics of antlers is crucially important to elucidate the clinical and medicinal efficacies of antlers. Hence, the review highlights information about various species of deer, its farming, antler preparation, antler composition, its traditional uses and scientific substantiation to it, dose and its future scope.
Courtesy source from:
Dr. Durgacharan Arun Bhagwat
Although rare now, some old recipes still call for Hartshorn. This product is made from distilled, ground antler by burning in kilns, which makes ammonium carbonate. This was the predecessor to baking powder, which is still used today in thin shaped cookies and crackers. When heated, the gaseous ammonia is released in the baking process, acting as a leveling agent.
Although you would not just bite into a shed antler or boil it up and put it on a plate, recently antlers have grown in popularity as dog chews. This is now a large part of the shed antler market. Dogs enjoy gnawing on them and get calcium as an added bonus.
ANTLER VELVET (Lu Rong)
IN CHINESE MEDICINE
IN CHINESE MEDICINE
The Chinese have utilized deer antler velvet as a Chinese Herb for thousands of years.
Today, usages of DAV include modern ailments such as high cholesterol, migraines, osteoporosis, muscle aches and pains, asthma, indigestion, cold hands and feet, disorders related to the liver and kidney, chronic skin sores or ulcers, overactive bladder, and sore or weak lower back and knees.
It has also been used to promote youthfulness, improve immunity, counter the effects of stress, speed the recovery from illness and injury, help the production and circulation of blood, protect the liver from toxins, and sharpen thinking skills.
Furthermore, the Chinese traditionally utilized Deer Antler Velvet as a tonic for children with developmental delays referred to as a “failure to thrive” resulting in slow growth, learning disabilities, and bone problems including rickets.
In Pinyin, deer antler velvet (or the velvet of young deer) is called Lu Rong which translates to Deer Antler (its pharmaceutical name is Cornu Cervi Parvum). It’s earliest known documentation as a Chinese Herb is in the Shen Nong Ben Cao Jing (Divine Husbandman’s Classic of Materia Medica). Researchers believe that this text is a compilation of oral traditions that were written down sometime between 200 and 250 CE.
CHEMICAL COMPOSITION : According to the text on Chinese Medical Herbology and Pharmacology,
chemically, Lu Rong is composed of : Pantocrine (Pantocrinum), lysophosphatidylcholine, ganglioside, putrescine, spermidine, spermine, PGE1, PGE2, PGF, chondroitin sulfate, androgen, estradiol, oestrone, ceramide, lecithin, cephalin, cholesterol, lipids, ganglioside, sphingomyelin, calcium phosphate, calcium carbonate, magnesium, and phosphorus.
Deer antler velvet is a Yang tonic that is sweet, salty and warm. It enters the Kidney and Liver channels. Traditional Chinese Theorists believe that because animals are closer to human beings than plants or other substances, animal substances are more beneficial than plants or minerals in tonifying the jing (essence) of the body. This idea of like treats like is referred to as the Doctrine of Similarity.
Furthermore, the ancient herbal text known as the Ben Cao Gang Mu (Materia Medica), expounds that deer are among the most helpful animals for tonifying yang and jing (essence). For this reason, many deer parts, including the horn/antler, blood, bone marrow, kidney, and placenta are used to tonify yang and replenish jing.
YIN AND YANG THEORY
What is all this discussion of yang, kidney, liver, and jing mean ?
In Chinese Theory, yin and yang describe the fundamental law of opposites that rule all phenomena. If something exists, it’s opposite or compliment must also exist to create balance. Opposite or contrary forces are considered complementary interconnected,
and interdependent in the natural world. They give rise to each other as they interrelate to one another. Day becomes night and night becomes day. Light and dark represent another tangible example of this duality, as do hot and cold or wet and dry.
As a general rule, Yin is the more passive, element associated with night, darkness, cold, water, the moon, heaviness, density, etc. Yang is the more active principle, and it supplies energy, light, the sun, fire, heat, etc. Nothing is ever wholly yin or yang; however, an imbalance in one will commonly create an imbalance in the opposite.
For example, if your car is running low on coolant, it will overheat because of a deficiency in yin (fluids) that give rise to an excess of heat. In Chinese Medicine, overheating due to a lack of fluids is known as Yin deficiency heat or Yin fire due to lack of fluids.
Deer antler velvet is contraindicated in cases of Yin fire because it is a warm herb in the same way that when a car overheats due to lack of fluids, the last thing you want to do is to expose it to more heat.
5-ELEMENT THEORY
From yin and yang theory, Chinese Medicine divides everything into 5-elements; wood, fire, earth, metal, and water.
Each of these five elements connects to the body’s physical organ systems and their corresponding meridians (or channels) that run through the body.
There are 12 major organ systems with related meridians and eight extra meridians that work on deeper channels of the body. Imagine that one of your organs, like the heart, is a lightbulb. The heart meridian is like an internal, energetic wiring system connected to that lightbulb that effects it and all its associated physical systems.
ZANG-FU PAIRS
Each of the 12 organs is a part of a Zang-Fu pair where the Zang organ is the yin organ, and the Fu organ is the yang organ. As an herb, deer antler velvet focuses on the Kidney and Liver organs. The Liver is a yin (Zang), wood organ, and the Kidney is a yin (Zang), water organ.
THE EIGHT EXTRAORDINARY VESSELS
Deer antler velvet also affects four of the eight extraordinary channels (vessels), including the Governing (Du Mai), Conception (Ren Mai), Penetrating (Chong Mai) and Girdle (Dai Mai) vessel, but delving into the extra channels is a bit advanced for this article.
YANG TONICS
Lu Rong (deer antler velvet) is considered a yang tonic herb. Most yang tonics focus on deficient yang of the Kidney, Heart, and Spleen, but the most significant use of the class of herbs focuses on Kidney Yang. Deficiency, in pinyin, is Xu (pronounced similarly to shoe), and you will more commonly hear Chinese Medicine practitioners refer to Kidney yang deficiency as Kidney yang xu.
Systemic Exhaustion is the principal manifestation of Kidney yang deficiency. Common signs and symptoms of Kidney yang xu include withdrawal from social interaction, a severe dislike or fear of cold, cold limbs, sore or weak lower back and knees, painful lower extremities, pale tongue, and a deep, weak pulse. Other problems frequently associated with Kidney yang xu include impotence, spermatorrhea (excessive, involuntary ejaculation), premature ejaculation, infertility in both males and females, watery vaginal discharge, enuresis (bed-wetting), polyuria (production of abnormally large volumes of dilute urine), wheezing, and daybreak diarrhea.
From a modern biomedical standpoint, Kidney yang xu is far from being understood. The endocrine system, which is a chemical messenger system consisting of a group of glands that produce and secrete hormones, appears to have a connection to a part of the etiology of Kidney Yang Xu.
From a biomedical standpoint, Yang Tonic herbs generally: Regulate the adrenal cortex which produces hormones that are vital to life, such as cortisol (which helps regulate metabolism and helps your body respond to stress) and aldosterone (which helps control blood pressure).Regulate energy metabolismPromote healthy sexual function and fertilityPromote growthStrengthen the body’s resistance to illness, degeneration, and injury
Because most yang tonics are warm and have drying effects, they can injure the yin and assist fire as explained by the earlier example of an overheating vehicle. For this reason, when patients have symptoms related to yin deficiency (xu) heat or fire, such as; thirst, dry mouth, night sweats, fevers, dizziness, vertigo, tinnitus, constipation, and loss of appetite yang tonics should be avoided.
JING
In Chinese Theory, the Kidneys store Jing. There is no direct translation for the Chinese word jing into English, so essence or “essential qi,” are often used. Consequently, there is no direct translation for qi into English either, are life-force or energy are considered the closest concepts.
Jing is considered to be the underpinning of all aspects of organic life. Protecting and preserving jing is instrumental for longevity. Stored in the kidneys, it is responsible for human growth, development, fertility, and many other functional activities. Two main kinds of jing exist, Prenatal Jing (Pre-Heaven Essence) and Postnatal Jing (Post-Heaven Essence).
The quantity of Prenatal Jing a person has is determined at birth when it passes onto the fetus from the parents. Prenatal Jing determines the constitution, strength, and vitality of the fetus. After that, Jing can only be conserved or used up more slowly. Living a balanced life of moderation in diet, work, rest and sexual activity may preserve Prenatal Jing. In Chinese Medicine, once all of the prenatal jing lost, we die.
Postnatal Jing is refined Qi from food, drink and practicing breathing exercises such as Qi Gong and Tai Chi.
Kidney Jing controls the growth of bones, teeth, hair, brain development, sexual maturation, reproductive function and fertility, and healthy development into adulthood. It declines naturally with age, producing the characteristic signs of hair or tooth loss, signs of aging, impairment of memory, etc. Kidney Jing also influences our strength and resistance, and if it is “wasted” or poorly stored, the person may have lowered immunity to exogenous pathogenic influences and suffer from constant illnesses. Deficiency of Kidney Jing also affects Kidney Qi in general and can result in problems like impotence, chronic weak or sore lower back, weak knees, tinnitus, urinary incontinence, deafness, loose teeth, etc.
DEER ANTLER VELVET IN DEPTH:
According to the Essentials of Chinese Medicine Materia Medica, deer antler velvet is “the ultimate tonic herb for the Liver and Kidney.” It tonifies the Governing Vessel (Du Mai) and stabilizes the Penetrating (Chong Mai) and Conception (Ren Mai) vessels, which are 3 of the extra meridians mentioned earlier.
Deer antler velvet’s ability to fortify Kidney yang, augment the essence and blood, and strengthen the sinews and bones make it a highly valued yang tonic in Chinese culture. Primary uses of deer antler velvet include Kidney yang deficiency, jing and blood deficiency, weakness of the sinews and bones, developmental delays in children, Gushing and Leaking Syndromes, excessive vaginal discharge due to unstable Penetrating (Chong Mai) and Conception (Ren Mai) vessels, and generating tissue to promote the healing of yin-type abscesses.
The four most significant Traditional Chinese Therapeutic uses of Deer Antler Velvet (Lu Rong).
TRADITIONAL CHINESE THERAPEUTIC ACTIONS
Tonifies Kidney Yang and Replenishes Kidney Jing (Essence)
Kidney Yang Deficiency with impotence: In Traditional Chinese Medicine (TCM), Lu Rong is a beneficial herb for Kidney yang and jing deficiencies.
Kidney yang and jing deficiency present with symptoms such as: fatigue, lassitude, lightheadedness, dizziness, tinnitus, impotence, premature ejaculation, spermatorrhea, nocturnal emissions, soreness or lack of strength of the lower back and knees, cold pain at the lower back and knees, cold extremities, weakness of sinews and joints, and frequent copious, clear urination.
Infertility, leukorrhea:
Another frequent use of deer antler velvet something known as insufficient Kidney fire resulting in deficiencies of the Ren (Conception) and Chong (Penetrating) channels. Clinical manifestations of depleted Kidney fire include clear, watery leukorrhea, constant uterine bleeding, and a cold womb which may lead to female infertility. Cold causes stagnation, which means that the body’s reproductive processes “freeze” up much in the same way that excess cold causes water to freeze and stop flowing naturally. Deer antler velvet warms the Kidney yang and the uterus, helping to regulate menstruation and contract the uterus to stop bleeding. It also treats deficient cold of the Ren (Conception) and Chong (Penetrating) channels with unremitting Gushing and Leaking syndrome and an unstable girdle (Dai Mai) vessel with a deficient cold type of excessive vaginal discharge.
Nourishes Blood, Ren (Conception) and Chong (Penetrating) Channels.
Severe anemia due to blood and Kidney Jing (essence) deficiencies:
Deer antler velvet is often used to counter severe anemia due to blood and Kidney Jing (essence) deficiencies. In Chinese Medicine, blood and jing arise from the same source. In cases of severe blood deficiency or blood loss, jing will be deficient as well. Deer antler velvet has traditionally been used to tonify blood and nourish jing to promote the production of red blood cells and reticulocytes (which are immature red blood cells). Therefore, in Chinese Medicine, deer antler velvet is believed to be well suited for patients that are thin and weak with severe blood and jing deficiencies.
Augments the Essence and Blood and Strengthens Sinews and Bones
Weakness of sinews and bones:
Chinese theory states that the Kidney stores jing (essence), which is vital for healthy bones, marrow, and a healthy brain. Furthermore, the Liver stores blood and controls the sinews and tendons. Thus, when the Liver and Kidney are deficient, the bones, sinews, and joints become weak. Deficiencies of Kidney yang and jing in children with lack of strength in sinews and bones can manifest as delayed mental and physical development. Deficiencies of Kidney yang and jing in elderly patients may lead to Wei (atrophy) syndrome, weakened bones, and joints, confusion, or memory loss.
Heals Chronic Yin Sores and Boils
In China, deer antler velvet has been used to help heal chronic non-healing ulcerations and sores or non-perforated yin-type ulcers or abscesses that are the result of qi and blood deficiency for hundreds of years. These sores are most commonly concave and discharge a clear fluid. They are also slow-healing in nature, with dark, dull skin surrounding the lesion.
PHARMACOLOGICAL EFFECTS
In the Chinese Medical Herbology and Pharmacology and the Chinese Herbal Medicine Materia Medica, a variety of pharmacological effects of Lu Rong have been analyzed from a biomedical perspective, including, but not limited to:
Cardiovascular:
In several animal studies it was shown that the cardiovascular effect of deer antler velvet varies according to dosage. With low doses, there was no significant change noted in the cardiovascular system. Moderate doses were shown to have a significant positive inotropic (modifying the force or speed of contraction of muscles) and chronotropic (affecting the heart rate) effect, leading to increased cardiac output in heart specimens. This effect was most significant when given to individuals with weak hearts. Furthermore, when it was given orally to subjects with chronic poor circulation and low blood pressure, it increased both the blood pressure and pulse strength. Large doses resulted in negative chronotropic and inotropic effects, leading to dilation of blood vessels (peripheral vasodilation) and a decrease in blood pressure. Its clinical applications for the cardiovascular system include treatment of arrhythmia and hypotension caused by excessive blood loss.
Endocrine:
In one study, mice received oral administration of deer antler velvet at the dosage of 100 to 200 mg/kg.
Researchers evaluated the plasma levels of testosterone before and after the dosage. Testosterone levels increased significantly in old and young mice, but there was no change observed in healthy adult mice. When rats that had their gonads removed received preparations of deer antler velvet, no significant sexual hormonal effect was noted.
Gastrointestinal:
Studies show that deer antler velvet stimulates the production of PGE2. PGE2, or prostaglandin E2, is a naturally occurring prostaglandin often used as a medication and as a principal mediator of inflammation. It has shown healing-promoting effects on gastric ulcers and intestinal lesions and is beneficial for patients with peptic ulcers.
Impact on local healing:
Pantocrine, one component of deer antler velvet, has been shown to enhance healing and promote granulation of chronic, long-standing wounds and ulcerations, as well as the healing of bone fractures. The local metabolism of nitrogen and carbohydrates is also affected by Pantocrine.
General strengthening:
Pantocrine, one component of deer antler velvet, is a general tonic that has demonstrated marked effectiveness to increase work capacity, improve sleep, increase appetite, and decrease the rate of muscle fatigue. When given deer antler velvet over an extended period, patients showed an increase in body weight and red blood cell count. In animal studies, deer antler velvet increased the oxygen uptake of the brain, liver, and kidneys of white rats. When rats ingested deer antler velvet, their body weight increased within two weeks.
Hematological effect:
When rabbits were given powdered deer antler velvet orally or injected with preparations of the substance, there was an increase in both red and white blood cells. When rabbits received large amounts of deer antler velvet, there was a marked increase in the production of red blood cells.Immunological effect: Pantocrinum given to animals that are sensitive to a particular substance inhibits a reaction when they are re-exposed to the material.
Impact on the Kidneys:
Preparations of deer antler velvet have a diuretic effect.
CLINICAL STUDIES AND RESEARCH
In the Chinese Medical Herbology and Pharmacology textbook, several studies on Lu Rong are presented, including:
Hyperplasia of mammary glands:
An injectable preparation of deer antler velvet showed 87.2% effectiveness in treating 86 women with hyperplasia of the mammary glands. The treatment protocol was to administer 2 ml via intramuscular injection, twice daily for 10 to 15 days before menstruation.
Diarrhea:
Patients with diarrhea characterized by deficient Kidney yang received one intramuscular injection of deer antler velvet daily, or every other day, for a total of two doses. Out of 16 patients, 12 had a complete recovery, 3 showed moderate improvement, and 1 showed no response.
Impotence:
Intramuscular injections of deer antler velvet given every other day and oral ingestion of herbs were administered daily to treat 42 patients with impotence, with marked effectiveness. The treatment protocol was to inject 0.5 ml of deer antler velvet into the acupuncture points Qihai (CV6), Guanyuan (CV 4), Zhongji (CV 3), Qugu (CV 2), and Zusanli (ST 36), and 1.0 ml into Mingmen (GV 4). The composition of the herbal formula varied depending on the presentation of each patient.Atrioventricular block: In one study, 20 patients with atrioventricular block were treated with 2.0 ml intramuscular injection of deer antler velvet for 25 to 30 days with an 85% effective rate.
ADDENDUM ON ANTLERS OF MATURE DEER:
Mature deer antlers are known as Lu Jiao (Cornu Cervi). This herb is salty and warm and enters the Liver and Kidney channels. The ability of Lu Jiao to tonify the Kidneys and assist the yang is weaker than that of deer antler velvet. However, Lu Jiao also invigorates blood and reduces toxic swelling and sores. As such it is used for breast abscesses, pain from blood stasis and deep pain in the lower back. The dosage of Lu Jiao is 5-10g either in decoctions or taken directly as a powder which can be ingested or applied topically.
Lu Jiao Jiao (Gelatinum Cornu Cervi), is a gelatin made from mature deer antlers. Lu Jiao Jiao is cheaper and weaker than deer antler velvet and is often substituted when the primary substance is not available. It is sweet, salty, slightly warm, and not as strong as the velvet in tonifying the Kidney Yang. However, it is still able to nourish and tonify essence and blood. It is also useful to stop bleeding. Lu Jiao Jiao is beneficial for patients with deficient and wasted jing and blood with bleeding or yin-type sores. The dosage is 6-12g.
The dregs left over from boiling Lu Jiao Jiao are known as Lu Jiao Shuang (Cornu Cervi Degelatinatium). Sweet, salty and slightly warm, Lu Jiao Shuang’s ability to augment jing and blood does not approach that of deer antler velvet. However, it does have a stronger retaining effect. Clinically, it is beneficial for deficient uterine bleeding and vaginal discharge due to cold. It can also be applied to stop bleeding.
Please note, all of these herbs are contraindicated in patients with heat from yin deficiency.
Info source
Velvet Antler: A Literature Review by Helen J. Batchelder, and Velvet Antler: A Summary of the Literature on health Benefits by Chris Tuckwell
DeerAntlerVelvet.com | Julius Cermak
Today, usages of DAV include modern ailments such as high cholesterol, migraines, osteoporosis, muscle aches and pains, asthma, indigestion, cold hands and feet, disorders related to the liver and kidney, chronic skin sores or ulcers, overactive bladder, and sore or weak lower back and knees.
It has also been used to promote youthfulness, improve immunity, counter the effects of stress, speed the recovery from illness and injury, help the production and circulation of blood, protect the liver from toxins, and sharpen thinking skills.
Furthermore, the Chinese traditionally utilized Deer Antler Velvet as a tonic for children with developmental delays referred to as a “failure to thrive” resulting in slow growth, learning disabilities, and bone problems including rickets.
In Pinyin, deer antler velvet (or the velvet of young deer) is called Lu Rong which translates to Deer Antler (its pharmaceutical name is Cornu Cervi Parvum). It’s earliest known documentation as a Chinese Herb is in the Shen Nong Ben Cao Jing (Divine Husbandman’s Classic of Materia Medica). Researchers believe that this text is a compilation of oral traditions that were written down sometime between 200 and 250 CE.
CHEMICAL COMPOSITION : According to the text on Chinese Medical Herbology and Pharmacology,
chemically, Lu Rong is composed of : Pantocrine (Pantocrinum), lysophosphatidylcholine, ganglioside, putrescine, spermidine, spermine, PGE1, PGE2, PGF, chondroitin sulfate, androgen, estradiol, oestrone, ceramide, lecithin, cephalin, cholesterol, lipids, ganglioside, sphingomyelin, calcium phosphate, calcium carbonate, magnesium, and phosphorus.
Deer antler velvet is a Yang tonic that is sweet, salty and warm. It enters the Kidney and Liver channels. Traditional Chinese Theorists believe that because animals are closer to human beings than plants or other substances, animal substances are more beneficial than plants or minerals in tonifying the jing (essence) of the body. This idea of like treats like is referred to as the Doctrine of Similarity.
Furthermore, the ancient herbal text known as the Ben Cao Gang Mu (Materia Medica), expounds that deer are among the most helpful animals for tonifying yang and jing (essence). For this reason, many deer parts, including the horn/antler, blood, bone marrow, kidney, and placenta are used to tonify yang and replenish jing.
YIN AND YANG THEORY
What is all this discussion of yang, kidney, liver, and jing mean ?
In Chinese Theory, yin and yang describe the fundamental law of opposites that rule all phenomena. If something exists, it’s opposite or compliment must also exist to create balance. Opposite or contrary forces are considered complementary interconnected,
and interdependent in the natural world. They give rise to each other as they interrelate to one another. Day becomes night and night becomes day. Light and dark represent another tangible example of this duality, as do hot and cold or wet and dry.
As a general rule, Yin is the more passive, element associated with night, darkness, cold, water, the moon, heaviness, density, etc. Yang is the more active principle, and it supplies energy, light, the sun, fire, heat, etc. Nothing is ever wholly yin or yang; however, an imbalance in one will commonly create an imbalance in the opposite.
For example, if your car is running low on coolant, it will overheat because of a deficiency in yin (fluids) that give rise to an excess of heat. In Chinese Medicine, overheating due to a lack of fluids is known as Yin deficiency heat or Yin fire due to lack of fluids.
Deer antler velvet is contraindicated in cases of Yin fire because it is a warm herb in the same way that when a car overheats due to lack of fluids, the last thing you want to do is to expose it to more heat.
5-ELEMENT THEORY
From yin and yang theory, Chinese Medicine divides everything into 5-elements; wood, fire, earth, metal, and water.
Each of these five elements connects to the body’s physical organ systems and their corresponding meridians (or channels) that run through the body.
There are 12 major organ systems with related meridians and eight extra meridians that work on deeper channels of the body. Imagine that one of your organs, like the heart, is a lightbulb. The heart meridian is like an internal, energetic wiring system connected to that lightbulb that effects it and all its associated physical systems.
ZANG-FU PAIRS
Each of the 12 organs is a part of a Zang-Fu pair where the Zang organ is the yin organ, and the Fu organ is the yang organ. As an herb, deer antler velvet focuses on the Kidney and Liver organs. The Liver is a yin (Zang), wood organ, and the Kidney is a yin (Zang), water organ.
THE EIGHT EXTRAORDINARY VESSELS
Deer antler velvet also affects four of the eight extraordinary channels (vessels), including the Governing (Du Mai), Conception (Ren Mai), Penetrating (Chong Mai) and Girdle (Dai Mai) vessel, but delving into the extra channels is a bit advanced for this article.
YANG TONICS
Lu Rong (deer antler velvet) is considered a yang tonic herb. Most yang tonics focus on deficient yang of the Kidney, Heart, and Spleen, but the most significant use of the class of herbs focuses on Kidney Yang. Deficiency, in pinyin, is Xu (pronounced similarly to shoe), and you will more commonly hear Chinese Medicine practitioners refer to Kidney yang deficiency as Kidney yang xu.
Systemic Exhaustion is the principal manifestation of Kidney yang deficiency. Common signs and symptoms of Kidney yang xu include withdrawal from social interaction, a severe dislike or fear of cold, cold limbs, sore or weak lower back and knees, painful lower extremities, pale tongue, and a deep, weak pulse. Other problems frequently associated with Kidney yang xu include impotence, spermatorrhea (excessive, involuntary ejaculation), premature ejaculation, infertility in both males and females, watery vaginal discharge, enuresis (bed-wetting), polyuria (production of abnormally large volumes of dilute urine), wheezing, and daybreak diarrhea.
From a modern biomedical standpoint, Kidney yang xu is far from being understood. The endocrine system, which is a chemical messenger system consisting of a group of glands that produce and secrete hormones, appears to have a connection to a part of the etiology of Kidney Yang Xu.
From a biomedical standpoint, Yang Tonic herbs generally: Regulate the adrenal cortex which produces hormones that are vital to life, such as cortisol (which helps regulate metabolism and helps your body respond to stress) and aldosterone (which helps control blood pressure).Regulate energy metabolismPromote healthy sexual function and fertilityPromote growthStrengthen the body’s resistance to illness, degeneration, and injury
Because most yang tonics are warm and have drying effects, they can injure the yin and assist fire as explained by the earlier example of an overheating vehicle. For this reason, when patients have symptoms related to yin deficiency (xu) heat or fire, such as; thirst, dry mouth, night sweats, fevers, dizziness, vertigo, tinnitus, constipation, and loss of appetite yang tonics should be avoided.
JING
In Chinese Theory, the Kidneys store Jing. There is no direct translation for the Chinese word jing into English, so essence or “essential qi,” are often used. Consequently, there is no direct translation for qi into English either, are life-force or energy are considered the closest concepts.
Jing is considered to be the underpinning of all aspects of organic life. Protecting and preserving jing is instrumental for longevity. Stored in the kidneys, it is responsible for human growth, development, fertility, and many other functional activities. Two main kinds of jing exist, Prenatal Jing (Pre-Heaven Essence) and Postnatal Jing (Post-Heaven Essence).
The quantity of Prenatal Jing a person has is determined at birth when it passes onto the fetus from the parents. Prenatal Jing determines the constitution, strength, and vitality of the fetus. After that, Jing can only be conserved or used up more slowly. Living a balanced life of moderation in diet, work, rest and sexual activity may preserve Prenatal Jing. In Chinese Medicine, once all of the prenatal jing lost, we die.
Postnatal Jing is refined Qi from food, drink and practicing breathing exercises such as Qi Gong and Tai Chi.
Kidney Jing controls the growth of bones, teeth, hair, brain development, sexual maturation, reproductive function and fertility, and healthy development into adulthood. It declines naturally with age, producing the characteristic signs of hair or tooth loss, signs of aging, impairment of memory, etc. Kidney Jing also influences our strength and resistance, and if it is “wasted” or poorly stored, the person may have lowered immunity to exogenous pathogenic influences and suffer from constant illnesses. Deficiency of Kidney Jing also affects Kidney Qi in general and can result in problems like impotence, chronic weak or sore lower back, weak knees, tinnitus, urinary incontinence, deafness, loose teeth, etc.
DEER ANTLER VELVET IN DEPTH:
According to the Essentials of Chinese Medicine Materia Medica, deer antler velvet is “the ultimate tonic herb for the Liver and Kidney.” It tonifies the Governing Vessel (Du Mai) and stabilizes the Penetrating (Chong Mai) and Conception (Ren Mai) vessels, which are 3 of the extra meridians mentioned earlier.
Deer antler velvet’s ability to fortify Kidney yang, augment the essence and blood, and strengthen the sinews and bones make it a highly valued yang tonic in Chinese culture. Primary uses of deer antler velvet include Kidney yang deficiency, jing and blood deficiency, weakness of the sinews and bones, developmental delays in children, Gushing and Leaking Syndromes, excessive vaginal discharge due to unstable Penetrating (Chong Mai) and Conception (Ren Mai) vessels, and generating tissue to promote the healing of yin-type abscesses.
The four most significant Traditional Chinese Therapeutic uses of Deer Antler Velvet (Lu Rong).
TRADITIONAL CHINESE THERAPEUTIC ACTIONS
Tonifies Kidney Yang and Replenishes Kidney Jing (Essence)
Kidney Yang Deficiency with impotence: In Traditional Chinese Medicine (TCM), Lu Rong is a beneficial herb for Kidney yang and jing deficiencies.
Kidney yang and jing deficiency present with symptoms such as: fatigue, lassitude, lightheadedness, dizziness, tinnitus, impotence, premature ejaculation, spermatorrhea, nocturnal emissions, soreness or lack of strength of the lower back and knees, cold pain at the lower back and knees, cold extremities, weakness of sinews and joints, and frequent copious, clear urination.
Infertility, leukorrhea:
Another frequent use of deer antler velvet something known as insufficient Kidney fire resulting in deficiencies of the Ren (Conception) and Chong (Penetrating) channels. Clinical manifestations of depleted Kidney fire include clear, watery leukorrhea, constant uterine bleeding, and a cold womb which may lead to female infertility. Cold causes stagnation, which means that the body’s reproductive processes “freeze” up much in the same way that excess cold causes water to freeze and stop flowing naturally. Deer antler velvet warms the Kidney yang and the uterus, helping to regulate menstruation and contract the uterus to stop bleeding. It also treats deficient cold of the Ren (Conception) and Chong (Penetrating) channels with unremitting Gushing and Leaking syndrome and an unstable girdle (Dai Mai) vessel with a deficient cold type of excessive vaginal discharge.
Nourishes Blood, Ren (Conception) and Chong (Penetrating) Channels.
Severe anemia due to blood and Kidney Jing (essence) deficiencies:
Deer antler velvet is often used to counter severe anemia due to blood and Kidney Jing (essence) deficiencies. In Chinese Medicine, blood and jing arise from the same source. In cases of severe blood deficiency or blood loss, jing will be deficient as well. Deer antler velvet has traditionally been used to tonify blood and nourish jing to promote the production of red blood cells and reticulocytes (which are immature red blood cells). Therefore, in Chinese Medicine, deer antler velvet is believed to be well suited for patients that are thin and weak with severe blood and jing deficiencies.
Augments the Essence and Blood and Strengthens Sinews and Bones
Weakness of sinews and bones:
Chinese theory states that the Kidney stores jing (essence), which is vital for healthy bones, marrow, and a healthy brain. Furthermore, the Liver stores blood and controls the sinews and tendons. Thus, when the Liver and Kidney are deficient, the bones, sinews, and joints become weak. Deficiencies of Kidney yang and jing in children with lack of strength in sinews and bones can manifest as delayed mental and physical development. Deficiencies of Kidney yang and jing in elderly patients may lead to Wei (atrophy) syndrome, weakened bones, and joints, confusion, or memory loss.
Heals Chronic Yin Sores and Boils
In China, deer antler velvet has been used to help heal chronic non-healing ulcerations and sores or non-perforated yin-type ulcers or abscesses that are the result of qi and blood deficiency for hundreds of years. These sores are most commonly concave and discharge a clear fluid. They are also slow-healing in nature, with dark, dull skin surrounding the lesion.
PHARMACOLOGICAL EFFECTS
In the Chinese Medical Herbology and Pharmacology and the Chinese Herbal Medicine Materia Medica, a variety of pharmacological effects of Lu Rong have been analyzed from a biomedical perspective, including, but not limited to:
Cardiovascular:
In several animal studies it was shown that the cardiovascular effect of deer antler velvet varies according to dosage. With low doses, there was no significant change noted in the cardiovascular system. Moderate doses were shown to have a significant positive inotropic (modifying the force or speed of contraction of muscles) and chronotropic (affecting the heart rate) effect, leading to increased cardiac output in heart specimens. This effect was most significant when given to individuals with weak hearts. Furthermore, when it was given orally to subjects with chronic poor circulation and low blood pressure, it increased both the blood pressure and pulse strength. Large doses resulted in negative chronotropic and inotropic effects, leading to dilation of blood vessels (peripheral vasodilation) and a decrease in blood pressure. Its clinical applications for the cardiovascular system include treatment of arrhythmia and hypotension caused by excessive blood loss.
Endocrine:
In one study, mice received oral administration of deer antler velvet at the dosage of 100 to 200 mg/kg.
Researchers evaluated the plasma levels of testosterone before and after the dosage. Testosterone levels increased significantly in old and young mice, but there was no change observed in healthy adult mice. When rats that had their gonads removed received preparations of deer antler velvet, no significant sexual hormonal effect was noted.
Gastrointestinal:
Studies show that deer antler velvet stimulates the production of PGE2. PGE2, or prostaglandin E2, is a naturally occurring prostaglandin often used as a medication and as a principal mediator of inflammation. It has shown healing-promoting effects on gastric ulcers and intestinal lesions and is beneficial for patients with peptic ulcers.
Impact on local healing:
Pantocrine, one component of deer antler velvet, has been shown to enhance healing and promote granulation of chronic, long-standing wounds and ulcerations, as well as the healing of bone fractures. The local metabolism of nitrogen and carbohydrates is also affected by Pantocrine.
General strengthening:
Pantocrine, one component of deer antler velvet, is a general tonic that has demonstrated marked effectiveness to increase work capacity, improve sleep, increase appetite, and decrease the rate of muscle fatigue. When given deer antler velvet over an extended period, patients showed an increase in body weight and red blood cell count. In animal studies, deer antler velvet increased the oxygen uptake of the brain, liver, and kidneys of white rats. When rats ingested deer antler velvet, their body weight increased within two weeks.
Hematological effect:
When rabbits were given powdered deer antler velvet orally or injected with preparations of the substance, there was an increase in both red and white blood cells. When rabbits received large amounts of deer antler velvet, there was a marked increase in the production of red blood cells.Immunological effect: Pantocrinum given to animals that are sensitive to a particular substance inhibits a reaction when they are re-exposed to the material.
Impact on the Kidneys:
Preparations of deer antler velvet have a diuretic effect.
CLINICAL STUDIES AND RESEARCH
In the Chinese Medical Herbology and Pharmacology textbook, several studies on Lu Rong are presented, including:
Hyperplasia of mammary glands:
An injectable preparation of deer antler velvet showed 87.2% effectiveness in treating 86 women with hyperplasia of the mammary glands. The treatment protocol was to administer 2 ml via intramuscular injection, twice daily for 10 to 15 days before menstruation.
Diarrhea:
Patients with diarrhea characterized by deficient Kidney yang received one intramuscular injection of deer antler velvet daily, or every other day, for a total of two doses. Out of 16 patients, 12 had a complete recovery, 3 showed moderate improvement, and 1 showed no response.
Impotence:
Intramuscular injections of deer antler velvet given every other day and oral ingestion of herbs were administered daily to treat 42 patients with impotence, with marked effectiveness. The treatment protocol was to inject 0.5 ml of deer antler velvet into the acupuncture points Qihai (CV6), Guanyuan (CV 4), Zhongji (CV 3), Qugu (CV 2), and Zusanli (ST 36), and 1.0 ml into Mingmen (GV 4). The composition of the herbal formula varied depending on the presentation of each patient.Atrioventricular block: In one study, 20 patients with atrioventricular block were treated with 2.0 ml intramuscular injection of deer antler velvet for 25 to 30 days with an 85% effective rate.
ADDENDUM ON ANTLERS OF MATURE DEER:
Mature deer antlers are known as Lu Jiao (Cornu Cervi). This herb is salty and warm and enters the Liver and Kidney channels. The ability of Lu Jiao to tonify the Kidneys and assist the yang is weaker than that of deer antler velvet. However, Lu Jiao also invigorates blood and reduces toxic swelling and sores. As such it is used for breast abscesses, pain from blood stasis and deep pain in the lower back. The dosage of Lu Jiao is 5-10g either in decoctions or taken directly as a powder which can be ingested or applied topically.
Lu Jiao Jiao (Gelatinum Cornu Cervi), is a gelatin made from mature deer antlers. Lu Jiao Jiao is cheaper and weaker than deer antler velvet and is often substituted when the primary substance is not available. It is sweet, salty, slightly warm, and not as strong as the velvet in tonifying the Kidney Yang. However, it is still able to nourish and tonify essence and blood. It is also useful to stop bleeding. Lu Jiao Jiao is beneficial for patients with deficient and wasted jing and blood with bleeding or yin-type sores. The dosage is 6-12g.
The dregs left over from boiling Lu Jiao Jiao are known as Lu Jiao Shuang (Cornu Cervi Degelatinatium). Sweet, salty and slightly warm, Lu Jiao Shuang’s ability to augment jing and blood does not approach that of deer antler velvet. However, it does have a stronger retaining effect. Clinically, it is beneficial for deficient uterine bleeding and vaginal discharge due to cold. It can also be applied to stop bleeding.
Please note, all of these herbs are contraindicated in patients with heat from yin deficiency.
Info source
Velvet Antler: A Literature Review by Helen J. Batchelder, and Velvet Antler: A Summary of the Literature on health Benefits by Chris Tuckwell
DeerAntlerVelvet.com | Julius Cermak
Lesser Mouse Deer 小鼷鹿
Tragulus kanchil
Tragulus kanchil
Shot at MT15 nite walk on Jan 2021
The lesser mouse-deer, lesser Malay chevrotain, or kanchil (Tragulus kanchil) is a species of even-toed ungulate in the family Tragulidae.
The lesser mouse-deer is found widely across Southeast Asia in Indochina, Burma (Kra Isthmus), Brunei, Cambodia, China (Southern Yunnan), Indonesia (Kalimantan, Sumatra and many other small islands), Laos, Malaysia (Peninsular Malaysia, Sarawak and many other small islands), Singapore, Thailand, and Vietnam.
It is the smallest known hoofed mammal, its mature size being as little as 45 cm (18 inches) and 2 kg (4.4 lb). It is threatened by predation by feral dogs.
The Malay or Indonesian name kancil, (pronounced 'kanchil' or 'kahn-cheel', as in the species' name) means both mouse-deer and 'clever person'. The generic name Tragulus, is composed of Tragos, 'goat' in Greek, and –ulus, meaning 'tiny' in Latin.
In an Indonesian and Malaysian folklore, the mouse-deer Sang Kancil is a cunning trickster similar to Br'er Rabbit from the Uncle Remus tales, even sharing some story plots, like when they both trick enemies pretending to be dead or inanimate, or lose a race to slower opponents.
Although the mousedeer in Southeast Asia resemble deer, they are distinguishable from the latter by their small size, thin legs, and a triangular white pattern extending from the chin and running down the throat.
Their coat ranges in colour from grey to reddish-brown. Males have a pair of enlarged canines that extend down from their upper jaw.
The head-to-body length of an adult greater mousedeer is about 50 to 60 cm, while that of an adult lesser mousedeer is around 40 to 50 cm.
The distinguishing feature between the two species lies in the white stripes on either side of the triangular pattern running down their chin and throat. The white stripes are continuous in the lesser mousedeer, but appear broken and/or uneven in the greater mousedeer.
The greater mousedeer has a gestation period of five to six months, and the young will reach full adult size at around five months old. The lesser mousedeer usually gives birth to one fawn, which is weaned at about three months old.
Mousedeer are frugivorous. They feed on low vegetation as well as fallen fruits, shoots, young leaves and fungi foraged from the ground.
The mousedeer inhabit both primary and mature secondary rainforests. Other than Singapore, both species can be found in Indochina, Peninsular Malaysia, Sumatra and Borneo. The lesser mousedeer is also found in southern China.
In Singapore, the lesser mousedeer has always been a known inhabitant of the Central Catchment Area. The greater mousedeer, on the other hand, was thought to have vanished from Singapore.
In 1999, eight greater mousedeer bred at the zoo were released into the Bukit Timah nature reserve and the MacRitchie Reservoir area.
In 2009, the National Parks Board confirmed an official sighting of the greater mousedeer at Pulau Ubin. Prior to that, there had also been sightings of both the lesser and greater mousedeer at Lower Peirce Reservoir since 2007.
The mousedeer is traditionally hunted for its meat, which is said to be more tender than venison. The meat is also made into dendeng (spiced, dried meat).
On 27 June 1947, the Wild Animals and Birds Protection Order, 1947, was gazetted, which put an end to the hunting of mousedeer and several other wildlife species in Singapore.
Phylum: Chordata
Class: Mammalia
Order: Artiodactyla
Family: Tragulidae
Genus: Tragulus
Species: T. kanchil
Binomial name Tragulus kanchil
Raffles, 1821
The lesser mouse-deer is found widely across Southeast Asia in Indochina, Burma (Kra Isthmus), Brunei, Cambodia, China (Southern Yunnan), Indonesia (Kalimantan, Sumatra and many other small islands), Laos, Malaysia (Peninsular Malaysia, Sarawak and many other small islands), Singapore, Thailand, and Vietnam.
It is the smallest known hoofed mammal, its mature size being as little as 45 cm (18 inches) and 2 kg (4.4 lb). It is threatened by predation by feral dogs.
The Malay or Indonesian name kancil, (pronounced 'kanchil' or 'kahn-cheel', as in the species' name) means both mouse-deer and 'clever person'. The generic name Tragulus, is composed of Tragos, 'goat' in Greek, and –ulus, meaning 'tiny' in Latin.
In an Indonesian and Malaysian folklore, the mouse-deer Sang Kancil is a cunning trickster similar to Br'er Rabbit from the Uncle Remus tales, even sharing some story plots, like when they both trick enemies pretending to be dead or inanimate, or lose a race to slower opponents.
Although the mousedeer in Southeast Asia resemble deer, they are distinguishable from the latter by their small size, thin legs, and a triangular white pattern extending from the chin and running down the throat.
Their coat ranges in colour from grey to reddish-brown. Males have a pair of enlarged canines that extend down from their upper jaw.
The head-to-body length of an adult greater mousedeer is about 50 to 60 cm, while that of an adult lesser mousedeer is around 40 to 50 cm.
The distinguishing feature between the two species lies in the white stripes on either side of the triangular pattern running down their chin and throat. The white stripes are continuous in the lesser mousedeer, but appear broken and/or uneven in the greater mousedeer.
The greater mousedeer has a gestation period of five to six months, and the young will reach full adult size at around five months old. The lesser mousedeer usually gives birth to one fawn, which is weaned at about three months old.
Mousedeer are frugivorous. They feed on low vegetation as well as fallen fruits, shoots, young leaves and fungi foraged from the ground.
The mousedeer inhabit both primary and mature secondary rainforests. Other than Singapore, both species can be found in Indochina, Peninsular Malaysia, Sumatra and Borneo. The lesser mousedeer is also found in southern China.
In Singapore, the lesser mousedeer has always been a known inhabitant of the Central Catchment Area. The greater mousedeer, on the other hand, was thought to have vanished from Singapore.
In 1999, eight greater mousedeer bred at the zoo were released into the Bukit Timah nature reserve and the MacRitchie Reservoir area.
In 2009, the National Parks Board confirmed an official sighting of the greater mousedeer at Pulau Ubin. Prior to that, there had also been sightings of both the lesser and greater mousedeer at Lower Peirce Reservoir since 2007.
The mousedeer is traditionally hunted for its meat, which is said to be more tender than venison. The meat is also made into dendeng (spiced, dried meat).
On 27 June 1947, the Wild Animals and Birds Protection Order, 1947, was gazetted, which put an end to the hunting of mousedeer and several other wildlife species in Singapore.
Phylum: Chordata
Class: Mammalia
Order: Artiodactyla
Family: Tragulidae
Genus: Tragulus
Species: T. kanchil
Binomial name Tragulus kanchil
Raffles, 1821
The banded pig (Sus scrofa vittatus)
The banded pig (Sus scrofa vittatus) also known as the Indonesian wild boar is a subspecies of wild boarnative to the Thai-Malay Peninsula and
many Indonesian islands, including Sumatra, Java, and the Lesser Sundas as far east as Komodo.
It is known as the wild boar in Singapore.
It is the most basal subspecies, having the smallest relative brain size, more primitive dentition, and unspecialised cranial structure. It is a short-faced subspecies with a white band on the muzzle, as well as sparse body hair, no underwool, a fairly long mane, and a broad reddish band extending from the muzzle to the sides of the neck. It is much smaller than the mainland S. s. cristatus subspecies, with the largest specimens on Komodo weighing only 48 kg.
In some areas, it differs from most other boar populations by being highly frugivorous, with specimens in Ujung Kulon National Park in Java eating around 50 different fruit species, especially figs, thus making them important seed dispersers. On the islands of Komodo and Rinca, its diet is more varied, encompassing roots, tubers, grasses, insects, fruits, snakes, and carrion. It also frequently eats crabs during low tide. Piglets are born from December to March in litters of two to six, and are raised in grass nests constructed by their mother. They are much less vividly striped than the young of S. s. scrofa.
Class: Mammalia
Order: Artiodactyla
Family: Suidae
Genus: Sus
Species: S. scrofa
Subspecies: S. s. vittatus
Trinomial name Sus scrofa vittatus Boie, 1828
many Indonesian islands, including Sumatra, Java, and the Lesser Sundas as far east as Komodo.
It is known as the wild boar in Singapore.
It is the most basal subspecies, having the smallest relative brain size, more primitive dentition, and unspecialised cranial structure. It is a short-faced subspecies with a white band on the muzzle, as well as sparse body hair, no underwool, a fairly long mane, and a broad reddish band extending from the muzzle to the sides of the neck. It is much smaller than the mainland S. s. cristatus subspecies, with the largest specimens on Komodo weighing only 48 kg.
In some areas, it differs from most other boar populations by being highly frugivorous, with specimens in Ujung Kulon National Park in Java eating around 50 different fruit species, especially figs, thus making them important seed dispersers. On the islands of Komodo and Rinca, its diet is more varied, encompassing roots, tubers, grasses, insects, fruits, snakes, and carrion. It also frequently eats crabs during low tide. Piglets are born from December to March in litters of two to six, and are raised in grass nests constructed by their mother. They are much less vividly striped than the young of S. s. scrofa.
Class: Mammalia
Order: Artiodactyla
Family: Suidae
Genus: Sus
Species: S. scrofa
Subspecies: S. s. vittatus
Trinomial name Sus scrofa vittatus Boie, 1828
Lesser short-nosed fruit bat
(Cynopterus brachyotis)
(Cynopterus brachyotis)
The lesser short-nosed fruit bat (Cynopterus brachyotis) is a species of megabat within the family Pteropodidae. It is a small bat that lives in South and Southeast Asia and Indonesia (Borneo). It weighs between 21 and 32 grams (0.74 and 1.13 oz), and/or 70 to 127 mm.[3] It occurs in many types of habitat, but most frequently in disturbed forest, including lower montane forest and tropical lowland rain forest, plus gardens, mangroves, and vegetation on beaches.
Lesser short-nosed fruit bats are generally brown to yellowish brown with a brighter collar. Adult males have dark orange collars whereas adult females have yellowish collars. An indistinct collar is observed in some immature bats. The edges of the ears and the wing bones are usually white. Individuals have two pairs of lower incisors, a fox-like face and large dark eyes. The head and body length is 7–8 cm (2.8–3.1 in), the forearm length is 6–7 cm (2.4–2.8 in), tail length is 0.8–1.0 cm (0.31–0.39 in), and ear length is 1.4–1.6 cm (0.55–0.63 in).
There are nine subspecies of lesser short-nosed fruit bat. Corbet and Hill listed 19 alternate names of
C. brachyotis, which include: Pachysoma brachyotis, P. duvaucelii, P. brevicaudatum, P. luzoniense,
C. grandidieri, C. marginatus var. scherzeri,
C. marginatus var. ceylonensis,
C. marginatus var. philippensis,
C. marginatus var. cuminggii,
C. marginatus var. andamanensis,
C. brachyoma, C. montanoi, C. minutus, C. minor,
C. babi, C. archipelagus and C. nusatenggara. Kitchener and Maharadatunkamsi considered luzoniensis and minutus as separate species while Hill and Thonglongya transferred angulatus to C. sphinx.
The lifespan of the lesser short-nosed fruit bat is approximately 20 to 30 years.
The greater short-nosed fruit bat is similar to the lesser short-nosed fruit bat but has generally longer forearms, longer ears and a much longer skull. P. lucasi has only one pair of lower incisors, a lack of white edges to the ears and a usually greyer color. C. horsfieldi is larger, with heavily cusped molars. M. ecaudatus usually has a more upturned nose, lacks a bright collar and tail, and has only one pair of lower incisors.
Lesser short-nosed fruit bats are frugivorous. They prefer aromatic fruit, especially mangoes. The bats feed mainly on small fruits by sucking out the juices and soft pulp. They also eat nectar and pollen.
Most pregnancies occur from March to June with peaks in January and September. Lactation corresponds with the peak of the rainy season as well as the fruiting season. Both sexes take care of the young. Males play an active role in lactation and feeding the young. They have mammary glands that are the same size as those of the female and exceed 8% of their overall body mass.
The mating system of lesser short-nosed fruit bats is polygynous. In the Philippines, most populations give birth twice a year and pregnant females have been found in almost all months. The period of gestation is approximately 3.5 to 4 months. The mother nurses the young with milk for about six to eight weeks. It takes about a year for the male to become sexually mature, and most females become pregnant at approximately six to eight months of age.
Lesser short-nosed fruit bats prefer to roost in small groups in trees, under leaves, and in caves. Young males may roost alone. It is common for one male to roost with up to four females. Females may gather in groups of up to 20. To feed, the bats bite off the center part of palm fruit clusters, leaving a hollow for hanging, which is also the method they use to construct a shelter. Males may spend more than two months chewing the veins of leaves and palm fronds until they fall to form a shelter. Individuals use tactile, visual, and acoustic stimuli to communicate. They forage with their acute sense of smell and navigate with their keen vision.
Lesser short-nosed fruit bats are free of terrestrial predation because of their ability to fly. Some human cultures consume them as a delicacy. They play important roles in plant pollination. Plants such as bananas, avocados, dates, mangoes and peaches, and depend on them for seed dispersal. The bats are considered to be crop pests since they consume and damage fruit.
Lesser short-nosed fruit bats are designated a least-concern species by the International Commission on Zoological Nomenclature (ICZN) because the population is widely distributed, stable and still abundant. Possible threats may be habitat loss due to development, dams, and deforestation. The animals are being hunted for medical purposes, as reported in ICZN 2006.
Class: Mammalia
Order: Chiroptera
Family: Pteropodidae
Genus: Cynopterus
Species: C. brachyotis
Binomial name Cynopterus brachyotis (Müller, 1838)
Lesser short-nosed fruit bats are generally brown to yellowish brown with a brighter collar. Adult males have dark orange collars whereas adult females have yellowish collars. An indistinct collar is observed in some immature bats. The edges of the ears and the wing bones are usually white. Individuals have two pairs of lower incisors, a fox-like face and large dark eyes. The head and body length is 7–8 cm (2.8–3.1 in), the forearm length is 6–7 cm (2.4–2.8 in), tail length is 0.8–1.0 cm (0.31–0.39 in), and ear length is 1.4–1.6 cm (0.55–0.63 in).
There are nine subspecies of lesser short-nosed fruit bat. Corbet and Hill listed 19 alternate names of
C. brachyotis, which include: Pachysoma brachyotis, P. duvaucelii, P. brevicaudatum, P. luzoniense,
C. grandidieri, C. marginatus var. scherzeri,
C. marginatus var. ceylonensis,
C. marginatus var. philippensis,
C. marginatus var. cuminggii,
C. marginatus var. andamanensis,
C. brachyoma, C. montanoi, C. minutus, C. minor,
C. babi, C. archipelagus and C. nusatenggara. Kitchener and Maharadatunkamsi considered luzoniensis and minutus as separate species while Hill and Thonglongya transferred angulatus to C. sphinx.
The lifespan of the lesser short-nosed fruit bat is approximately 20 to 30 years.
The greater short-nosed fruit bat is similar to the lesser short-nosed fruit bat but has generally longer forearms, longer ears and a much longer skull. P. lucasi has only one pair of lower incisors, a lack of white edges to the ears and a usually greyer color. C. horsfieldi is larger, with heavily cusped molars. M. ecaudatus usually has a more upturned nose, lacks a bright collar and tail, and has only one pair of lower incisors.
Lesser short-nosed fruit bats are frugivorous. They prefer aromatic fruit, especially mangoes. The bats feed mainly on small fruits by sucking out the juices and soft pulp. They also eat nectar and pollen.
Most pregnancies occur from March to June with peaks in January and September. Lactation corresponds with the peak of the rainy season as well as the fruiting season. Both sexes take care of the young. Males play an active role in lactation and feeding the young. They have mammary glands that are the same size as those of the female and exceed 8% of their overall body mass.
The mating system of lesser short-nosed fruit bats is polygynous. In the Philippines, most populations give birth twice a year and pregnant females have been found in almost all months. The period of gestation is approximately 3.5 to 4 months. The mother nurses the young with milk for about six to eight weeks. It takes about a year for the male to become sexually mature, and most females become pregnant at approximately six to eight months of age.
Lesser short-nosed fruit bats prefer to roost in small groups in trees, under leaves, and in caves. Young males may roost alone. It is common for one male to roost with up to four females. Females may gather in groups of up to 20. To feed, the bats bite off the center part of palm fruit clusters, leaving a hollow for hanging, which is also the method they use to construct a shelter. Males may spend more than two months chewing the veins of leaves and palm fronds until they fall to form a shelter. Individuals use tactile, visual, and acoustic stimuli to communicate. They forage with their acute sense of smell and navigate with their keen vision.
Lesser short-nosed fruit bats are free of terrestrial predation because of their ability to fly. Some human cultures consume them as a delicacy. They play important roles in plant pollination. Plants such as bananas, avocados, dates, mangoes and peaches, and depend on them for seed dispersal. The bats are considered to be crop pests since they consume and damage fruit.
Lesser short-nosed fruit bats are designated a least-concern species by the International Commission on Zoological Nomenclature (ICZN) because the population is widely distributed, stable and still abundant. Possible threats may be habitat loss due to development, dams, and deforestation. The animals are being hunted for medical purposes, as reported in ICZN 2006.
Class: Mammalia
Order: Chiroptera
Family: Pteropodidae
Genus: Cynopterus
Species: C. brachyotis
Binomial name Cynopterus brachyotis (Müller, 1838)
Finlayson's squirrel or the Variable squirrel
Finlayson's squirrel or the variable squirrel is a species of rodent in the family Sciuridae.
It is found in Cambodia, Laos, Myanmar, Thailand, and Vietnam. The species occurs in a wide range of wooded habitats, including gardens and parks in cities like Bangkok.
Scientific name: Callosciurus finlaysonii
Conservation status: Least Concern
(Population stable) Encyclopedia of Life
Rank: Species
Higher classification: Oriental Tree Squirrels
Phylum: Chordata
Class: Mammalia
It is found in Cambodia, Laos, Myanmar, Thailand, and Vietnam. The species occurs in a wide range of wooded habitats, including gardens and parks in cities like Bangkok.
Scientific name: Callosciurus finlaysonii
Conservation status: Least Concern
(Population stable) Encyclopedia of Life
Rank: Species
Higher classification: Oriental Tree Squirrels
Phylum: Chordata
Class: Mammalia
Slender Squirrel
The slender squirrel (Sundasciurus tenuis) is a species of rodent in the Sciuridae family.
It is arboreal and found in Indonesia, Singapore, Malaysia, and Thailand. The body is brown on the upper parts and light grey on the underparts.
The body measures about 13–16 cm, with a slightly shorter slender tail. It feeds on soft bark, fruits and insects.
Scientific name: Sundasciurus tenuis
Mass: 75 g (Large Adult) Encyclopedia of Life
Conservation status: Least Concern (Population decreasing) Encyclopedia of Life
Trophic level: Omnivorous Encyclopedia of Life
Phylum: Chordata
Length: 17 cm (Large Adult) Encyclopedia of Life
It is arboreal and found in Indonesia, Singapore, Malaysia, and Thailand. The body is brown on the upper parts and light grey on the underparts.
The body measures about 13–16 cm, with a slightly shorter slender tail. It feeds on soft bark, fruits and insects.
Scientific name: Sundasciurus tenuis
Mass: 75 g (Large Adult) Encyclopedia of Life
Conservation status: Least Concern (Population decreasing) Encyclopedia of Life
Trophic level: Omnivorous Encyclopedia of Life
Phylum: Chordata
Length: 17 cm (Large Adult) Encyclopedia of Life
Plantain Squirrel
The plantain squirrel, oriental squirrel or tricoloured squirrel is a species of rodents in the family Sciuridae found in Indonesia, Malaysia, Singapore, and Thailand in a wide range of habitats: forests, mangroves, parks, gardens, and agricultural areas. Fruit farmers consider them to be pests.
Its body is about 20–30 cm (8–12 in) long with a similar-sized tail. It is greyish/brown with a chestnut belly and a black and white stripe on the side.
The genus name Callosciurus means beautiful squirrels. Its diet consists mostly of leaves and fruits, but it also eats insects and bird eggs. It is known to break open twigs that contain ant larvae to eat them.
It can eat fruits much bigger than itself, such as mangoes, jackfruit, or coconuts. It is very quick and agile in trees, able to jump a few metres between trees, and rarely wanders on the ground.
Scientific name: Callosciurus notatus
Mass: 190 g (Large Adult) Encyclopedia of Life
Trophic level: Omnivorous Encyclopedia of Life
Conservation status: Least Concern (Population increasing) Encyclopedia of Life
Length: 20 cm (Large Adult) Encyclopedia of Life
Rank: Species
Its body is about 20–30 cm (8–12 in) long with a similar-sized tail. It is greyish/brown with a chestnut belly and a black and white stripe on the side.
The genus name Callosciurus means beautiful squirrels. Its diet consists mostly of leaves and fruits, but it also eats insects and bird eggs. It is known to break open twigs that contain ant larvae to eat them.
It can eat fruits much bigger than itself, such as mangoes, jackfruit, or coconuts. It is very quick and agile in trees, able to jump a few metres between trees, and rarely wanders on the ground.
Scientific name: Callosciurus notatus
Mass: 190 g (Large Adult) Encyclopedia of Life
Trophic level: Omnivorous Encyclopedia of Life
Conservation status: Least Concern (Population increasing) Encyclopedia of Life
Length: 20 cm (Large Adult) Encyclopedia of Life
Rank: Species
Sunda Flying Lemur 飞狐猴
Galeopterus variegatus
Galeopterus variegatus
The Sunda flying lemur (Galeopterus variegatus) or Sunda colugo, also known as the Malayan flying lemur or Malayan colugo, is a species of colugo.
Until recently, it was thought to be one of only two species of flying lemur, the other being the Philippine flying lemur which is found only in the Philippines. The Sunda flying lemur is found throughout Southeast Asia in Indonesia, Thailand, Malaysia, and Singapore.
The Sunda flying lemur is not a lemur and does not fly. Instead, it glides as it leaps among trees. It is strictly arboreal, is active at night, and feeds on soft plant parts such as young leaves, shoots, flowers, and fruits. After a 60-day gestation period, a single offspring is carried on the mother's abdomen held by a large skin membrane. It is a forest-dependent species.
The head-body length of Sunda flying lemur is about 33 to 42 cm (13 to 17 in). Its tail length measures 18 to 27 cm (7.1 to 10.6 in),[5] and its weight is 0.9 to 1.3 kg (2.0 to 2.9 lb).
The Sunda flying lemur is protected by national legislation. In addition to deforestation and loss of habitat, local subsistence hunting poses a serious threat to this animal. Competition with the plantain squirrel (Callosciurus notatus) represents another challenge for this species. More information is needed on population declines, but at present, the rate of the decline is not believed to merit listing in any category lower than Least Concern.
Habits and lifestyle
Sunda flying lemurs live either solitary or in small groups that are loosely connected. They can be territorial as regards foraging and sleeping areas. They are mainly nocturnal. They are strictly arboreal and in the daytime, they sleep high within dense foliage in the treetops or in holes in trees. With all four of their feet, they cling on to the trunk of a tree or the underside of branches. Climbing involves stretching out their two front legs and then bringing up their two back legs, which results in an awkward hopping. They can glide more than 100 m with minimal loss in elevation. When threatened they either climb higher up or remain motionless.
Behaviour and ecology
The Sunda flying lemur is a skillful climber, but is helpless when on the ground. Its gliding membrane connects from the neck, extending along the limbs to the tips of the fingers, toes and nails. This kite-shaped skin is known as a patagium, which is expanded for gliding. It can glide over a distance of 100 m with a loss of fewer than 10 m in elevation.
It can maneuver and navigate while gliding, but strong rain and wind can affect its ability to glide. Gliding usually occurs in open areas or high in the canopy, especially in dense tropical rainforest. The Sunda flying lemur needs a certain distance to glide and to land to avoid injury. The highest landing forces are experienced after short glides; longer glides lead to softer landings, due to the colugo's ability to break its glide aerodynamically. The ability to glide increases a colugo's access to scattered food resources in the rainforest, without increasing exposure to terrestrial or arboreal predators.
In general, the diet of the Sunda flying lemur consists mainly of leaves. It usually consumes leaves with less potassium and nitrogen-containing compounds, but with higher tannin. It also feeds on buds, shoots, coconut flowers, durian flowers, fruits, and sap from selected tree species. It also feeds on insects in Sarawak, Malaysian Borneo. The selected food sources depend on the localities, habitat, vegetation types, and availability.
The Sunda flying lemur mainly forages in tree canopies. It may forage on several different tree species in a single night or on a single species. It can also be seen licking tree bark of selected tree species to obtain water, nutrients, salts, and minerals. Though the Sunda flying lemur has been reported to also occur in gardens and plantations, the species resides in forests primarily.
Distributions and habitats
The Sunda flying lemur is widely distributed throughout Southeast Asia, ranging from the Sunda Shelf mainland to other islands – Northern Laos, Cambodia, Vietnam, Thailand, Malaysia (Peninsular, Sabah and Sarawak), Singapore, Brunei, Indonesia (Kalimantan, Sumatra, Bali, Java) and many adjacent islands. Conversely, the Philippine flying lemur
(C. volans) is confined to the southern parts of the Philippines only.
The Sunda flying lemur is adapted to many different vegetation types, including gardens, primary and secondary forest, rubber and coconut plantations, fruit orchards (dusun), mangrove swamps, lowlands and upland forests, tree plantations, lowland dipterocarp forests, and mountainous areas. However, not all of the mentioned habitats can sustain large colugo populations.
Scientific classification
Kingdom: Animalia
Phylum: Chordata
Class: Mammalia
Order: Dermoptera
Family: Cynocephalidae
Genus: Galeopterus Thomas, 1908
Species: G. variegatus
Binomial name Galeopterus variegatus
Until recently, it was thought to be one of only two species of flying lemur, the other being the Philippine flying lemur which is found only in the Philippines. The Sunda flying lemur is found throughout Southeast Asia in Indonesia, Thailand, Malaysia, and Singapore.
The Sunda flying lemur is not a lemur and does not fly. Instead, it glides as it leaps among trees. It is strictly arboreal, is active at night, and feeds on soft plant parts such as young leaves, shoots, flowers, and fruits. After a 60-day gestation period, a single offspring is carried on the mother's abdomen held by a large skin membrane. It is a forest-dependent species.
The head-body length of Sunda flying lemur is about 33 to 42 cm (13 to 17 in). Its tail length measures 18 to 27 cm (7.1 to 10.6 in),[5] and its weight is 0.9 to 1.3 kg (2.0 to 2.9 lb).
The Sunda flying lemur is protected by national legislation. In addition to deforestation and loss of habitat, local subsistence hunting poses a serious threat to this animal. Competition with the plantain squirrel (Callosciurus notatus) represents another challenge for this species. More information is needed on population declines, but at present, the rate of the decline is not believed to merit listing in any category lower than Least Concern.
Habits and lifestyle
Sunda flying lemurs live either solitary or in small groups that are loosely connected. They can be territorial as regards foraging and sleeping areas. They are mainly nocturnal. They are strictly arboreal and in the daytime, they sleep high within dense foliage in the treetops or in holes in trees. With all four of their feet, they cling on to the trunk of a tree or the underside of branches. Climbing involves stretching out their two front legs and then bringing up their two back legs, which results in an awkward hopping. They can glide more than 100 m with minimal loss in elevation. When threatened they either climb higher up or remain motionless.
Behaviour and ecology
The Sunda flying lemur is a skillful climber, but is helpless when on the ground. Its gliding membrane connects from the neck, extending along the limbs to the tips of the fingers, toes and nails. This kite-shaped skin is known as a patagium, which is expanded for gliding. It can glide over a distance of 100 m with a loss of fewer than 10 m in elevation.
It can maneuver and navigate while gliding, but strong rain and wind can affect its ability to glide. Gliding usually occurs in open areas or high in the canopy, especially in dense tropical rainforest. The Sunda flying lemur needs a certain distance to glide and to land to avoid injury. The highest landing forces are experienced after short glides; longer glides lead to softer landings, due to the colugo's ability to break its glide aerodynamically. The ability to glide increases a colugo's access to scattered food resources in the rainforest, without increasing exposure to terrestrial or arboreal predators.
In general, the diet of the Sunda flying lemur consists mainly of leaves. It usually consumes leaves with less potassium and nitrogen-containing compounds, but with higher tannin. It also feeds on buds, shoots, coconut flowers, durian flowers, fruits, and sap from selected tree species. It also feeds on insects in Sarawak, Malaysian Borneo. The selected food sources depend on the localities, habitat, vegetation types, and availability.
The Sunda flying lemur mainly forages in tree canopies. It may forage on several different tree species in a single night or on a single species. It can also be seen licking tree bark of selected tree species to obtain water, nutrients, salts, and minerals. Though the Sunda flying lemur has been reported to also occur in gardens and plantations, the species resides in forests primarily.
Distributions and habitats
The Sunda flying lemur is widely distributed throughout Southeast Asia, ranging from the Sunda Shelf mainland to other islands – Northern Laos, Cambodia, Vietnam, Thailand, Malaysia (Peninsular, Sabah and Sarawak), Singapore, Brunei, Indonesia (Kalimantan, Sumatra, Bali, Java) and many adjacent islands. Conversely, the Philippine flying lemur
(C. volans) is confined to the southern parts of the Philippines only.
The Sunda flying lemur is adapted to many different vegetation types, including gardens, primary and secondary forest, rubber and coconut plantations, fruit orchards (dusun), mangrove swamps, lowlands and upland forests, tree plantations, lowland dipterocarp forests, and mountainous areas. However, not all of the mentioned habitats can sustain large colugo populations.
Scientific classification
Kingdom: Animalia
Phylum: Chordata
Class: Mammalia
Order: Dermoptera
Family: Cynocephalidae
Genus: Galeopterus Thomas, 1908
Species: G. variegatus
Binomial name Galeopterus variegatus
Common Treeshrew 树鼩
Tupaia glis
Tupaia glis
The common treeshrew (Tupaia glis) is a small mammal in the treeshrew family Tupaiidae, and is native to Thailand, Malaysia, and Indonesia. It has been listed as Least Concern by IUCN as it remains common and displays some adaptability to ongoing habitat loss.
The common treeshrew is one of the largest among treeshrews. Average body length is between 16 and 21 cm (6.3 and 8.3 in), and average weight is around 190 g, with varying colours of reddish-brown, greyish or black upper parts and whitish belly.
Its long, bushy tail is dark greyish-brown and almost reaches the length of the body. The paws are bare with sharp nails, and with a naked patch of skin above its long nose. Both sexes are similar. The animal has a head and body length of 13–21 cm and a tail length of 12–20 cm. The common treeshrew usually has a white, pale stripe on each shoulder.
The two subspecies are T. g. longipes and T. g. salatana, with T. g. longipes being duller in color than T. g. salatana. The underparts of T. g. longipes are dull buff to reddish-buff, and the underside of the tail is greyish. The underparts and underside of the tail are dark reddish in T. g. salatana. Similar species are Tupaia splendidula and Tupaia montana.
Common treeshrews occur south of about 10°N latitude in southern Thailand through mainland Malaysia and adjacent coastal islands to Singapore.
They inhabit protected areas, including the Pasoh Forest Reserve on the Malay Peninsula and Krau Wildlife Reserve. In Indonesia, they are found on the islands of Siberut, Batu, Sumatra, Java, Bangka, Riau, Lingga and Anambas. Usually they are found in primary dipterocarp forest, but are tolerant to some degree of habitat modification. They have also been recorded from secondary forest, plantations, fruit orchards, and trees near housing areas.
Common treeshrews are probably present throughout the lowlands and hills up to 1,100 m (3,600 ft) in the Kelabit Highlands of Borneo. The subspecies T. g. longipes occurs in the north of Borneo, in Sarawak, and in East Kalimantan, including Sabah. The subspecies T. g. salatana occurs in the south of Rajang River and Kayan River in Borneo.
Common treeshrews are active during the day, and forage for food alone or in pairs, mainly on the ground, among shrubs and tree holes. They feed on fruits, seeds, leaves, and insects, especially ants and spiders. They are also reported to catch lizards.
They are very agile in climbing both large vertical tree trunks and bushes, and occasionally jump from stems of a young tree to that of another as much as 60 cm (24 in) away. Their climbing is concentrated in lower heights.
They frequently scent-mark their territories by chest and anogenital rubbing with a secretion from glands on chest and scrotum. Adult males are more secretory than females and juveniles. In the Bukit Timah Nature Reserve, mean home ranges of adult males were estimated at 10,174 m2 (109,510 sq ft), of adult females at 8,809 m2 (94,820 sq ft), of juvenile males at 7,527 m2 (81,020 sq ft), and of juvenile females at 7,255 m2 (78,090 sq ft), with partial overlaps between male and female ranges varying from 0.4% to 56.8%.
Home ranges of adult residents of the same sex overlap to a lesser degree than those of opposite sexes. A male's range may include the ranges of two or three females. A high overlap between ranges of one adult male and one adult female indicates they form a stable pair. Juvenile ranges of either sex adjoin or overlap with ranges of adults, suggesting the juveniles are family members. Individuals of the same sex are involved in aggressive territorial chases.
Juvenile males depart from their family's territory sooner than juvenile females.
Both sexes of common treeshrews are sexually mature at the age of about three months. In captivity, females give birth for the first time at the age of about 4.5 mo, usually in February. A postpartum oestrus results in more births in April.
Their oestrus cycle is eight to 39 days, and the gestation period lasts 40 to 52 days, after which a litter of one to three individuals is born.
The newborn offspring weigh about 10 to 12 grams. Females suckle their young every other day, and neglect their young as long as possible. They would not even be able to identify their own young if they did not mark them with the scent produced from glands in their sternum and abdomen.
Juveniles leave the nest between 25 and 35 days of age. Longevity of a captive common treeshrew has been recorded as 12 yr and 5 mo.
From October to December, common treeshrews are reproductively inactive. The mating season starts at the onset of the monsoon season in December and lasts until February.
Oestrus and preoestrus behavior is characterized by adult males pursuing adult females. Males emit chattering, and appear to be extremely excited. They also chase each other and fight. Females do not actively choose a partner among the male participants of chases. The dominant male gains access to females.
In tropical rainforest habitats in West Malaysia, population density varies from two to five animals per hectare. Their annual breeding coincides with the abundance of invertebrates after the dry season.
Their main reproductive period is between February and June, and their litter size is invariably two. Some females breed more than once a season, and the age at first pregnancy is seven months. The main period of emigration or mortality of young is during the breeding period or monsoon.
Common treeshrews are threatened due to deforestation and ensuing human activities in agriculture (for example, using trenchers to dig ditches), plantations, and commercial logging. Moreover, other pressures, such as hunting for food and sport, can create pressure to the species.
As a model organism
Tupaia glis is used by researchers as animal models for human diseases because of their close relationship to primates, and their well-developed senses of vision and hearing. Research studies have included hepatitis. Another instance has been documented where an individual of Tupaia glis developed breast cancer.
Order: Scandentia
Family: Tupaiidae
Genus: Tupaia
Species: T. glis
Binomial name Tupaia glis
The common treeshrew is one of the largest among treeshrews. Average body length is between 16 and 21 cm (6.3 and 8.3 in), and average weight is around 190 g, with varying colours of reddish-brown, greyish or black upper parts and whitish belly.
Its long, bushy tail is dark greyish-brown and almost reaches the length of the body. The paws are bare with sharp nails, and with a naked patch of skin above its long nose. Both sexes are similar. The animal has a head and body length of 13–21 cm and a tail length of 12–20 cm. The common treeshrew usually has a white, pale stripe on each shoulder.
The two subspecies are T. g. longipes and T. g. salatana, with T. g. longipes being duller in color than T. g. salatana. The underparts of T. g. longipes are dull buff to reddish-buff, and the underside of the tail is greyish. The underparts and underside of the tail are dark reddish in T. g. salatana. Similar species are Tupaia splendidula and Tupaia montana.
Common treeshrews occur south of about 10°N latitude in southern Thailand through mainland Malaysia and adjacent coastal islands to Singapore.
They inhabit protected areas, including the Pasoh Forest Reserve on the Malay Peninsula and Krau Wildlife Reserve. In Indonesia, they are found on the islands of Siberut, Batu, Sumatra, Java, Bangka, Riau, Lingga and Anambas. Usually they are found in primary dipterocarp forest, but are tolerant to some degree of habitat modification. They have also been recorded from secondary forest, plantations, fruit orchards, and trees near housing areas.
Common treeshrews are probably present throughout the lowlands and hills up to 1,100 m (3,600 ft) in the Kelabit Highlands of Borneo. The subspecies T. g. longipes occurs in the north of Borneo, in Sarawak, and in East Kalimantan, including Sabah. The subspecies T. g. salatana occurs in the south of Rajang River and Kayan River in Borneo.
Common treeshrews are active during the day, and forage for food alone or in pairs, mainly on the ground, among shrubs and tree holes. They feed on fruits, seeds, leaves, and insects, especially ants and spiders. They are also reported to catch lizards.
They are very agile in climbing both large vertical tree trunks and bushes, and occasionally jump from stems of a young tree to that of another as much as 60 cm (24 in) away. Their climbing is concentrated in lower heights.
They frequently scent-mark their territories by chest and anogenital rubbing with a secretion from glands on chest and scrotum. Adult males are more secretory than females and juveniles. In the Bukit Timah Nature Reserve, mean home ranges of adult males were estimated at 10,174 m2 (109,510 sq ft), of adult females at 8,809 m2 (94,820 sq ft), of juvenile males at 7,527 m2 (81,020 sq ft), and of juvenile females at 7,255 m2 (78,090 sq ft), with partial overlaps between male and female ranges varying from 0.4% to 56.8%.
Home ranges of adult residents of the same sex overlap to a lesser degree than those of opposite sexes. A male's range may include the ranges of two or three females. A high overlap between ranges of one adult male and one adult female indicates they form a stable pair. Juvenile ranges of either sex adjoin or overlap with ranges of adults, suggesting the juveniles are family members. Individuals of the same sex are involved in aggressive territorial chases.
Juvenile males depart from their family's territory sooner than juvenile females.
Both sexes of common treeshrews are sexually mature at the age of about three months. In captivity, females give birth for the first time at the age of about 4.5 mo, usually in February. A postpartum oestrus results in more births in April.
Their oestrus cycle is eight to 39 days, and the gestation period lasts 40 to 52 days, after which a litter of one to three individuals is born.
The newborn offspring weigh about 10 to 12 grams. Females suckle their young every other day, and neglect their young as long as possible. They would not even be able to identify their own young if they did not mark them with the scent produced from glands in their sternum and abdomen.
Juveniles leave the nest between 25 and 35 days of age. Longevity of a captive common treeshrew has been recorded as 12 yr and 5 mo.
From October to December, common treeshrews are reproductively inactive. The mating season starts at the onset of the monsoon season in December and lasts until February.
Oestrus and preoestrus behavior is characterized by adult males pursuing adult females. Males emit chattering, and appear to be extremely excited. They also chase each other and fight. Females do not actively choose a partner among the male participants of chases. The dominant male gains access to females.
In tropical rainforest habitats in West Malaysia, population density varies from two to five animals per hectare. Their annual breeding coincides with the abundance of invertebrates after the dry season.
Their main reproductive period is between February and June, and their litter size is invariably two. Some females breed more than once a season, and the age at first pregnancy is seven months. The main period of emigration or mortality of young is during the breeding period or monsoon.
Common treeshrews are threatened due to deforestation and ensuing human activities in agriculture (for example, using trenchers to dig ditches), plantations, and commercial logging. Moreover, other pressures, such as hunting for food and sport, can create pressure to the species.
As a model organism
Tupaia glis is used by researchers as animal models for human diseases because of their close relationship to primates, and their well-developed senses of vision and hearing. Research studies have included hepatitis. Another instance has been documented where an individual of Tupaia glis developed breast cancer.
Order: Scandentia
Family: Tupaiidae
Genus: Tupaia
Species: T. glis
Binomial name Tupaia glis